We study and orchestrate how synthetic materials interact with living cells. We engineer cell-instructive environments and material-based solutions for zero-waste therapeutic solutions
Our group develops hydrogel materials with programmed and tunable properties designed to encapsulate and instruct living cells. We study how living cells and inert matter interact and how these interactions can be exploited to direct cellular functions and ultimately result in therapeutic advantages. We cooperate with synthetic biologists, biophysicists, drug developers and clinicians to explore the application potential of our developments, with a focus on new materials for ophthalmic drug delivery. We contribute to INM’s competence fields opto-interactive and bio-intelligent materials. Our research addresses biomedical needs.

Kontakt
Mitarbeiter/innen






Forschung
Hydrogels with latent properties
By integrating phototriggers and light-responsive molecular motors in polymeric networks, we develop 4D hydrogels with optoregulated (bio)chemical activity, crosslinking, degradation, or mechanoactuation. These are used for biophysical studies of cell response to changes in the biochemical and mechanical signals of the extracellular environment.
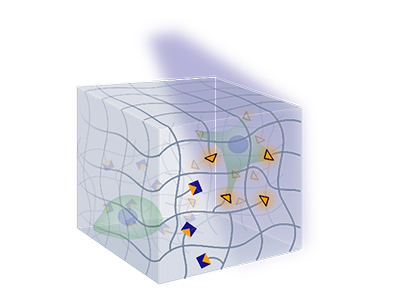
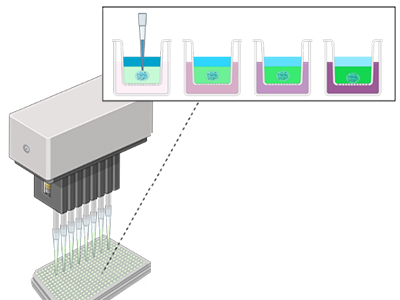
Model Cellular Microenvironments
We build synthetic models of cell-matrix and cell-cell interfaces with encoded biochemical, mechanical and dimensional signals. To accelerate discovery, we develop material microarrays for high-throughput biophysical experimentation and apply them to study multifactorial cell responses.
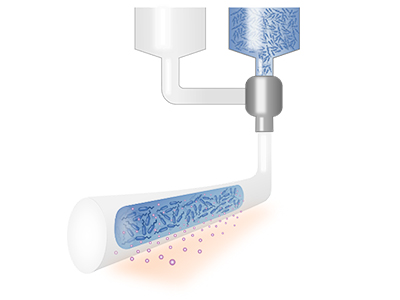
Living Therapeutic Devices
We develop bioinks and apply biofabrication technologies for functional and safe encapsulation of cells in medical devices. We focus on self-replenishable living therapeutic materials which integrate drug biofactories and have unlimited therapeutic release. We aim for innovation in ocular therapeutics with living, self-replenishable drug-eluting contact lenses.
Partner

Publikationen
Puertas-Bartolomé, María | Gutierrez-Urrutia, Izabook | Teruel-Enrico, Lara Luana | Dong, Nguyen Cao | Desai, Krupansh K. | Trujillo, Sara | Wittmann, Christoph | del Campo, Aránzazu
DOI:
The increasing prevalence of dry eye syndrome in aging and digital societies compromises long-term contact lens (CL) wear and forces users to regular eye drop instillation to alleviate discomfort. Here a novel approach with the potential to improve and extend the lubrication properties of CLs is presented. This is achieved by embedding lubricant-secreting biofactories within the CL material. The self-replenishable reservoirs autonomously produce and release hyaluronic acid (HA), a natural lubrication and wetting agent, long term. The hydrogel matrix regulates the growth of the biofactories and the HA production, and allows the diffusion of nutrients and HA for at least 3 weeks. The continuous release of HA sustainably reduces the friction coefficient of the CL surface. A self-lubricating CL prototype is presented, where the functional biofactories are contained in a functional ring at the lens periphery, outside of the vision area. The device is cytocompatible and fulfils physicochemical requirements of commercial CLs. The fabrication process is compatible with current manufacturing processes of CLs for vision correction. It is envisioned that the durable-by-design approach in living CL could enable long-term wear comfort for CL users and minimize the need for lubricating eye drops.
Milos, Frano | Del Campo, Aránzazu
DOI:
Polyacrylamide (PAAm) hydrogels are widely adopted as 2D-model soft substrates for investigating cell-material interactions in a controlled in vitro environment. They offer facile synthesis, tunable physico-chemical properties, diverse biofunctionalization routes, optical transparency, mouldability in a range of geometries and shapes, and compatibility with living cells. PAAm hydrogels can be engineered to reconstruct physiologically relevant biointerfaces, like cell-matrix or cell–cell interfaces, featuring biochemical, mechanical, and topographical cues present in the extracellular environment. This Review provides a materials science perspective on PAAm material properties, fabrication, and modification strategies relevant to cell studies, highlighting their versatility and potential to address a wide range of biological questions. Current routes are presented to integrate cell-instructive features, such as 2D patterns, 2.5D surface topographies, or mechanical stiffness gradients. Finally, the recent advances are emphasized toward dynamic PAAm hydrogels with on-demand control over hydrogel properties as well as electrically conductive PAAm hydrogels for bioelectronics.
Bhusari, Shardul | Kim, Juhyun | Polizzi, Karen | Sankaran, Shrikrishnan | del Campo, Aránzazu
DOI:
In engineered living materials (ELMs) non-living matrices encapsulate microorganisms to acquire capabilities like sensing or biosynthesis. The confinement of the organisms to the matrix and the prevention of overgrowth and escape during the lifetime of the material is necessary for the application of ELMs into real devices. In this study, a bilayer thin film hydrogel of Pluronic F127 and Pluronic F127 acrylate polymers supported on a solid substrate is introduced. The inner hydrogel layer contains genetically engineered bacteria and supports their growth, while the outer layer acts as an envelope and does not allow leakage of the living organisms outside of the film for at least 15 days. Due to the flat and transparent nature of the construct, the thin layer is suited for microscopy and spectroscopy-based analyses. The composition and properties of the inner and outer layer are adjusted independently to fulfil viability and confinement requirements. We demonstrate that bacterial growth and light-induced protein production are possible in the inner layer and their extent is influenced by the crosslinking degree of the used hydrogel. Bacteria inside the hydrogel are viable long term, they can act as lactate-sensors and remain active after storage in phosphate buffer at room temperature for at least 3 weeks. The versatility of bilayer bacteria thin-films is attractive for fundamental studies and for the development of application-oriented ELMs.
Yanamandra, Archana K. | Bhusari, Shardul | del Campo, Aránzazu | Sankaran, Shrikrishnan | Qu, Bin
DOI:
In living therapeutic materials (LTMs), organisms genetically programmed to produce and deliver drugs are encapsulated in porous matrices acting as physical barriers between the therapeutic organisms and the host cells. LTMs consisting of engineered E. coli encapsulated in Pluronic F127-based hydrogels have been frequently used in LTM designs but their immunogenicity has not been tested. In this study, we investigate the response of human peripheral blood mononuclear cells (PBMCs) exposed to this bacteria/hydrogel combination. The release of inflammation-related cytokines and cytotoxic proteins and the subsets of natural killer cells and T cells were examined. Encapsulation of the bacteria in hydrogels considerably lowers their immunogenicity. ClearColi, an endotoxin-free variant of E. coli, did not polarize NK cells into the more cytolytic CD16dim subset as E. coli. Our results demonstrate that ClearColi-encapsulated hydrogels generate low immunogenic response and are suitable candidates for the development of LTMs for in vivo testing to assess a potential clinical use. Nevertheless, we observed a stronger immune response (elevated levels of IFNγ, IL-6 and cytotoxic proteins) in pro-inflammatory PBMCs characterized by a high spontaneous release of IL-2. This highlights the need to identify recipients who have a higher likelihood of experiencing undesired immune responses to LTMs with IL-2 serving as a potential predictive marker. Additionally, including anti-inflammatory measures in living therapeutic material designs could be beneficial for such recipients.
Miguel-Jiménez, Adrián de | Ebeling, Bastian | Paez, Julieta I | Fink-Straube, Claudia | Pearson, Samuel | del Campo, Aránzazu
DOI:
Hydrogel precursors that crosslink within minutes are essential for the development of cell encapsulation matrices and their implementation in automated systems. Such timescales allow sufficient mixing of cells and hydrogel precursors under low shear forces and the achievement of homogeneous networks and cell distributions in the 3D cell culture. The previous work showed that the thiol-tetrazole methylsulfone (TzMS) reaction crosslinks star-poly(ethylene glycol) (PEG) hydrogels within minutes at around physiological pH and can be accelerated or slowed down with small pH changes. The resulting hydrogels are cytocompatible and stable in cell culture conditions. Here, the gelation kinetics and mechanical properties of PEG-based hydrogels formed by thiol-TzMS crosslinking as a function of buffer, crosslinker structure and degree of TzMS functionality are reported. Crosslinkers of different architecture, length and chemical nature (PEG versus peptide) are tested, and degree of TzMS functionality is modified by inclusion of RGD cell-adhesive ligand, all at concentration ranges typically used in cell culture. These studies corroborate that thiol/PEG-4TzMS hydrogels show gelation times and stiffnesses that are suitable for 3D cell encapsulation and tunable through changes in hydrogel composition. The results of this study guide formulation of encapsulating hydrogels for manual and automated 3D cell culture.
Riedel, Florian | Puertas Bartolomé, María | Teruel Enrico, Lara Luana | Fink-Straube, Claudia | Nguyen Dong, Cao | Gherlone, Fabio | Huang, Ying | Valiante, Vito | del Campo, Aránzazu | Sankaran, Shrikrishnan
DOI:
Microbial biofactories allow the upscaled production of high-value compounds in biotechnological processes. This is particularly advantageous for compounds like flavonoids that promote better health through their antioxidant, antibacterial, anticancer and other beneficial effects but are only produced in small quantities in their natural plant-based hosts. Bacteria like E. coli have been genetically modified with enzyme cascades to produce flavonoids like naringenin and pinocembrin from coumaric or cinnamic acid. Despite advancements in yield optimization, the production of these compounds still involves high costs associated with their biosynthesis, purification, storage and transport. An alternative production strategy could involve the direct delivery of the microbial biofactories to the body. In such a strategy, ensuring biocontainment of the engineered microbes in the body and controlling production rates are major challenges. In this study, these two aspects are addressed by developing engineered living materials (ELMs) consisting of probiotic microbial biofactories encapsulated in biocompatible hydrogels. Engineered probiotic E. coli Nissle 1917 able to efficiently convert cinnamic acid into pinocembrin were encapsulated in poly(vinyl alcohol)-based hydrogels. The biofactories are contained in the hydrogels for a month and remain metabolically active during this time. Control over production levels is achieved by the containment inside the material, which regulates bacteria growth, and by the amount of cinnamic acid in the medium.
Trujillo, Sara | Kasper, Jennifer Y. | Miguel-Jimenez, Adrián De | Abt, Britta | Bauer, Alina | Mekontso Ngaffo, Joelle A. | Pearson, Samuel | del Campo, Aránzazu
DOI:
Methylsulfone derivatized poly(ethylene) glycol (PEG) macromers can be biofunctionalized with thiolated ligands and cross-linked with thiol-based cross-linkers to obtain bioactive PEG hydrogels for in situ cell encapsulation. Methylsulfonyl-thiol (MS-SH) reactions present several advantages for this purpose when compared to other thiol-based cross-linking systems. They proceed with adequate and tunable kinetics for encapsulation, they reach a high conversion degree with good selectivity, and they generate stable reaction products. Our previous work demonstrated the cytocompatibility of cross-linked PEG-MS/thiol hydrogels in contact with fibroblasts. However, the cytocompatibility of the in situ MS-SH cross-linking reaction itself, which generates methylsulfinic acid as byproduct at the cross-linked site, remains to be evaluated. These studies are necessary to evaluate the potential of these systems for in vivo applications. Here we perform an extensive cytocompatibility study of PEG hydrogels during in situ cross-linking by the methylsulfonyl-thiol reaction. We compare these results with maleimide–thiol cross-linked PEGs which are well established for cell culture and in vivo experiments and do not involve the release of a byproduct. We show that fibroblasts and endothelial cells remain viable after in situ polymerization of methylsulfonyl-thiol gels on the top of the cell layers. Cell viability seems better than after in situ cross-linking hydrogels with maleimide–thiol chemistry. The endothelial cell proinflammatory phenotype is low and similar to the one obtained by the maleimide–thiol reaction. Finally, no activation of monocytes is observed. All in all, these results demonstrate that the methylsulfonyl-thiol chemistry is cytocompatible and does not trigger high pro-inflammatory responses in endothelial cells and monocytes. These results make methylsulfonyl-thiol chemistries eligible for in vivo testing and eventually clinical application in the future.
Vandeberg, Romain | Grysan, Patrick | Sion, Caroline | Włodarczyk-Biegun, Małgorzata K. | Lentzen, Esther | Bour, Jérôme | Krishnamoorthy, Sivashankar | Olmos, Eric | Grandfils, Christian
DOI:
AbstractSeveral clinical studies have reported the benefit of the administration of Mesenchymal Stem Cells (MSCs) in cell therapies. However, their routine applications need new substrates to amplify MSCs in vitro according to Good Manufacturing Practices (GMP) conditions and microcarriers are particularly suited for these purposes. In order to optimize the surface properties of Cytodex I microcarriers (Cyt), poly N-isopropylacrylamide (pNIPAM) has been grafted on their surface to promote MSCs adhesion, proliferation, but also to control their detachment by a decrease in temperature. The polymer coating generated on the microcarriers was analyzed using Time-of-Flight, Nanoscale Secondary Ion Mass Spectrometry, and Atomic Force Microscopy. We have confirmed the success of the pNIPAM grafting on Cyt with a two-steps reaction and correlated the influence on matrix functionalization in the function of the organic solvent used to disperse the microcarriers. The effects of pNIPAM functionalization have been explored on Wharton?s jelly-MSCs (WJ-MSCs) culture and cell thermal detachment was monitored with fluorescent microscopy. The in vitro results have indicated that WJ-MSCs have a better growth on Cyt-pNIPAM. However, pNIPAM thermal cell detachment was lower than trypsinization, implying that the minimum effective molecular weight and surface density of polymer chains have still to be future optimized.
Li, Bin | Çolak, Arzu | Blass, Johanna | Han, Mitchell | Zhang, Jingnan | Zheng, Yijun | Jiang, Qiyang | Bennewitz, Roland | Campo, Aránzazu del
DOI:
Understanding cells' response to the macroscopic and nanoscale properties of biomaterials requires studies in model systems with the possibility to tailor their mechanical properties and different length scales. Here, we describe an interpenetrating network (IPN) design based on a stiff PEGDA host network interlaced within a soft 4-arm PEG-Maleimide/thiol (guest) network. We quantify the nano- and bulk mechanical behavior of the IPN and the single network hydrogels by single-molecule force spectroscopy and rheological measurements. The IPN presents different mechanical cues at the molecular scale, depending on which network is linked to the probe, but the same mechanical properties at the macroscopic length scale as the individual host network. Cells attached to the interpenetrating (guest) network of the IPN or to the single network (SN) PEGDA hydrogel modified with RGD adhesive ligands showed comparable attachment and spreading areas, but cells attached to the guest network of the IPN, with lower molecular stiffness, showed a larger number and size of focal adhesion complexes and a higher concentration of the Hippo pathway effector Yes-associated protein (YAP) than cells linked to the PEGDA single network. The observations indicate that cell adhesion to the IPN hydrogel through the network with lower molecular stiffness proceeds effectively as if a higher ligand density is offered. We claim that IPNs can be used to decipher how changes in ECM design and connectivity at the local scale affect the fate of cells cultured on biomaterials.

Bhusari, Shardul | Sankaran, Shrikrishnan | del Campo, Aránzazu
DOI:
Engineered living materials (ELMs) are a new class of materials in which living organism incorporated into diffusive matrices uptake a fundamental role in material's composition and function. Understanding how the spatial confinement in 3D can regulate the behavior of the embedded cells is crucial to design and predict ELM's function, minimize their environmental impact and facilitate their translation into applied materials. This study investigates the growth and metabolic activity of bacteria within an associative hydrogel network (Pluronic-based) with mechanical properties that can be tuned by introducing a variable degree of acrylate crosslinks. Individual bacteria distributed in the hydrogel matrix at low density form functional colonies whose size is controlled by the extent of permanent crosslinks. With increasing stiffness and elastic response to deformation of the matrix, a decrease in colony volumes and an increase in their sphericity are observed. Protein production follows a different pattern with higher production yields occurring in networks with intermediate permanent crosslinking degrees. These results demonstrate that matrix design can be used to control and regulate the composition and function of ELMs containing microorganisms. Interestingly, design parameters for matrices to regulate bacteria behavior show similarities to those elucidated for 3D culture of mammalian cells.



