We study and orchestrate how synthetic materials interact with living cells. We engineer cell-instructive environments and material-based solutions for zero-waste therapeutic solutions
Our group develops hydrogel materials with programmed and tunable properties designed to encapsulate and instruct living cells. We study how living cells and inert matter interact and how these interactions can be exploited to direct cellular functions and ultimately result in therapeutic advantages. We cooperate with synthetic biologists, biophysicists, drug developers and clinicians to explore the application potential of our developments, with a focus on new materials for ophthalmic drug delivery. We contribute to INM’s competence fields opto-interactive and bio-intelligent materials. Our research addresses biomedical needs.

Kontakt
Mitarbeiter/innen






Forschung
Hydrogels with latent properties
By integrating phototriggers and light-responsive molecular motors in polymeric networks, we develop 4D hydrogels with optoregulated (bio)chemical activity, crosslinking, degradation, or mechanoactuation. These are used for biophysical studies of cell response to changes in the biochemical and mechanical signals of the extracellular environment.
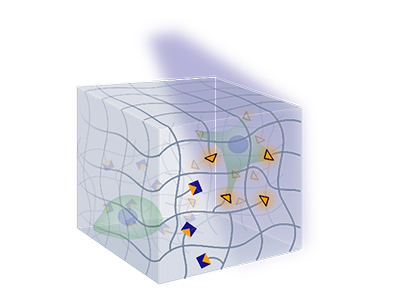
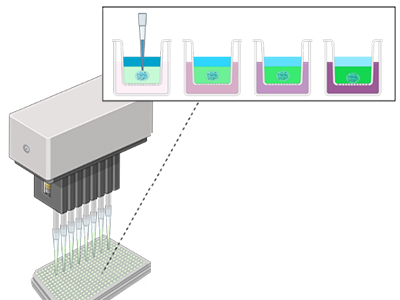
Model Cellular Microenvironments
We build synthetic models of cell-matrix and cell-cell interfaces with encoded biochemical, mechanical and dimensional signals. To accelerate discovery, we develop material microarrays for high-throughput biophysical experimentation and apply them to study multifactorial cell responses.
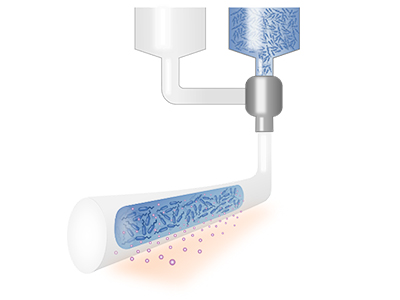
Living Therapeutic Devices
We develop bioinks and apply biofabrication technologies for functional and safe encapsulation of cells in medical devices. We focus on self-replenishable living therapeutic materials which integrate drug biofactories and have unlimited therapeutic release. We aim for innovation in ocular therapeutics with living, self-replenishable drug-eluting contact lenses.
Partner

Publikationen
Hambardzumyan, Syuzanna | Kasper, Jennifer Y. | Del Campo, Aránzazu
DOI:
Collagen matrix deposition is an important biomarker to predict the regenerative capacity of new biomaterials or the therapeutic potential of new drugs in collagen-associated diseases. Several methods for the quantification of matrix collagen in tissue samples are established, e.g., Picro-Sirius red assay, hydroxyproline assay, antibody-based assays, or the 3,4-DHPAA-based assay. These methods have been extended to quantify deposited collagen in in vitro cell culture models, although their applicability has been questioned due to the much lower concentration and eventually lower relative abundance of deposited collagen in cell cultures than in tissue. Here we compare the performance of the above-mentioned methods for the quantification of deposited matrix collagen in 2D cell cultures under different conditions: culture time, addition of collagen deposition-stimulating molecules, and post-culture processing step (decellularization). We show that the available methods can deliver accurate results within different experimental windows. We provide a comprehensive analysis of the relevant experimental parameters that influence the assay, and the sensitivity limits for the different methods, as well as the involved effort. In a comparative table, we provide guidance for the selection of the most appropriate collagen quantification assay for different culture conditions.
Liu, Kunpeng | Nickl, Philip | Feng, Jun | Haag, Rainer
DOI:
Blood-contacting medical devices play a crucial role in clinical interventions, but their susceptibility to thrombosis and inflammation poses serious risks to treatment outcomes and patient safety. This study presents a novel coating that combines dendritic polyglycerol amine (dPGA), dendritic polyglycerol aldehyde (dPG-CHO), and linear polyglycerol sulfate (lPGS) using a layer-by-layer self-assembly method (LBL) on a polystyrene surface. The immobilization of dendritic polyglycerol enhances surface coverage, enabling the incorporation of a higher density of heparin-mimicking lPGS, while the covalent bonding ensures the coating's long-term stability. Compared to the pristine substrate, the coating significantly reduced platelet adhesion and activation. Notably, its hemocompatibility effects persist even after 30 days. Furthermore, co-incubation experiments with RAW264.7 macrophages confirmed the anti-inflammatory properties of the polyglycerol-based coating. These results demonstrate that this heparin-mimetic coating effectively improves the hemocompatibility of polystyrene and has the potential to be applied to other blood-contacting materials.
Desai, Krupansh | Garcia-Sanchez, Lorely | Amini, Maryam | Teruel Enrico, Lara Luana | Siegrist, Silke | Del Campo, Aránzazu | Trujillo, Sara
DOI:
In ophthalmology, living biomaterials have appeared as promising drug delivery and biosensor devices to tackle dynamic sensing and delivery of compounds. Their living character complicates their assessment with the also dynamic ocular tissues. The use of animal experiments increases complexity, and most animal ocular models are anatomically different from humans. Thus, in vitro ocular systems applied specifically to living biomaterials are required to assess their safety, compatibility and efficacy. Here, we report on an in vitro cornea model for co-cultures with Corynebacterium glutamicum-polyvinyl alcohol living biomaterials, which are reported as suitable living contact lenses, to study their cytocompatibility to the eye. We co-cultured this living biomaterial with human primary corneal cells (epithelial and fibroblasts) for 7 days, mimicking contact lens extended wear. We studied bacterial proliferation, biocontainment and biosafety. We investigated potential cytotoxicity and pro-inflammatory responses of living biomaterials to corneal cells. Our results revealed that the living biomaterial does not trigger cytotoxicity or pro-inflammatory phenotypes on corneal cells during the 7-day co-culture. We placed the living biomaterial on top of the corneal epithelium, observing no cytotoxic effects. Overall, these findings highlight the potential of in vitro investigations for living biomaterials and the applicability of these devices for ophthalmology purposes.
Kafrashian, Zahra | Feng, Jun | Del Campo, Aránzazu
DOI:
Multimaterial optical fibers provide a versatile platform for integrating diverse functionalities—such as waveguiding, side emission, sensing, and actuation—into a single filament. Although traditional multimaterial fibers have primarily been fabricated from rigid materials such as silica and thermoplastics for optoelectronic applications, recent developments have shifted the focus toward soft systems composed of elastomers, hydrogels, and their composites. Owing to their mechanical compliance and biocompatibility, these soft fibers are particularly well suited for wearable, implantable, and tissue-integrated devices used in diagnostics and phototherapy. This review provides a comprehensive overview of the rapidly developing field of soft multimaterial optical fibers, highlighting key material combinations and fabrication strategies that enable multifunctional performance. Particular emphasis is placed on extrusion-based multimaterial printing—including coaxial and segmented extrusion—which has significantly expanded the architectural and functional design space of soft optical fibers. Remaining challenges, including material compatibility, interfacial and surface quality, and printing resolution, are critically discussed. Finally, the review outlines emerging opportunities for advancing these fabrication approaches toward practical and clinically relevant biomedical applications.
Kumar, Rinku | Fernandez-Yague, Marc A. | Bessaguet, Adrien | Lee, Hosoowi | Giuseppone, Nicolas | García, Andrés J. | Del Campo, Aránzazu
DOI:
Cellular force sensing and transduction are fundamental processes in development, homeostasis, and disease. To understand how cells detect and integrate mechanical forces, we need non-invasive methods to apply forces at the molecular scale while monitoring cellular responses within physiological contexts. Here, we present a mechanoactuated hydrogel interface that can exert forces on integrin adhesion receptors and allows monitoring of traction force responses in real time. The actuation is achieved by light excitation of a rotary molecular motor presenting an adhesion peptide to bind integrins at the cell membrane and to a hydrogel surface via flexible polymer chains. Illumination results in chain twisting and an applied pulling force on the linked integrin receptors within subcellular illuminated areas. Fluorescent particles in the hydrogel allow parallel quantification of cellular forces by traction force microscopy. With this methodology, we monitored talin recruitment, actin organization, and traction force generation and their reversibility in response to applied forces by the rotary motor-interface. We demonstrate reversible talin recruitment, enhanced F-actin polymerization, and a reduction in cell traction force when force is applied to focal adhesions. This research expands the application of nano machine-based actuation within soft hydrogels and showcases its capabilities.
Fernandes-Yagüe, Marc A. | Barber, Graham | del Campo, Aránzazu | Garcia, Andres J.
DOI:
Fibrotic capsule formation remains a major barrier in the clinical performance of biomedical implants. Here, we demonstrate that synthetic hydrogels mimicking the mechanical properties of fibrotic tissue trigger stromal cell activation and immune remodeling via focal adhesion kinase (FAK)-mediated mechanotransduction. Using a mechanically tunable poly(ethylene glycol) hydrogel platform and subcutaneous implantation in mice, we show that pharmacological inhibition of FAK activity significantly reduces α-smooth muscle actin (α-SMA)-positive myofibroblast activation, collagen I deposition, and fibrotic capsule thickness in a hydrogel stiffness-dependent manner. Flow cytometry and cytokine profiling revealed that FAK inhibition alters the fibrotic niche by reducing CD163-positive M2c macrophages and significantly downregulating pro-fibrotic cytokines including IL-6, and VEGF, while transiently increasing regulatory T cells and elevating IL-10 levels. Importantly, these changes occurred without parallel increases in canonical pro-inflammatory cytokines, indicating selective modulation rather than global immune suppression or activation. These findings position FAK as a central hub translating mechanical cues into coordinated stromal and immune responses. Targeting FAK mechanotransduction may provide a therapeutic strategy to mitigate foreign body responses and improve implant integration across regenerative applications.
Zhao, Renping | Zhang, Jingnan | Zhang, Sijia | Schwarz, Eva C. | Del Campo, Aránzazu | Hoth, Markus | Qu, Bin
DOI:
T cell activation requires cell polarization and changes in gene expression. Target cell stiffness contributes to the activation of immune cells, and tumor cell softening is linked to cancer progression. We investigated how substrate stiffness influences T cell activation using functionalized, T cell–activating substrates of varying stiffness and softened target cells. Reorientation of the microtubule-organizing center (MTOC) toward the immunological synapse and nuclear translocation of the transcription factor NFAT1 were impaired on softer hydrogels or upon contact with softer target cells. The increase in intracellular Ca2+ induced by target engagement also depended on stiffness and was reduced on soft substrates. Stiffness-dependent Ca2+ signaling was crucial for both rapid (MTOC reorientation) and long-term (NFAT translocation) responses. Whereas MTOC reorientation depended on the mechanosensitive Ca2+-permeable channel PIEZO1, NFAT1 translocation depended on the Ca2+ channel ORAI1. Our results demonstrate that target stiffness directly influences MTOC reorientation and NFAT1 translocation in T cells, and these two processes are governed by different plasma membrane Ca2+ channels, indicating that these stiffness-regulated rapid and long-term responses can be decoupled. Our findings imply that tumor cell stiffness regulates T cell functionality and suggest that pathways regulated by PIEZO1 and ORAI1 might differentially control rapid and long-term responses to stiffness in other cell types.
Farrukh, Hafiz Syed Usama Bin | Farrukh, Aleeza | Hambardzumyan, Syuzanna | Steudter, Therese | Del Campo, Aránzazu
DOI:
Hydrogels mimicking the mechanical and biochemical features of the cellular microenvironment allow cell encapsulation and facilitate in vitro 3D culture. In addition to biocompatibility and reactivity in physiological conditions, a key criterion for crosslinking chemistry is appropriate gelation kinetics to allow mixing and homogeneous distribution of cells with the hydrogel precursors. We have previously presented aryl methylsulfone/thiol (MS/SH) reaction as a thiol-reactive cross-linking system for cell encapsulation in star polyethylene glycol (PEG4) hydrogels with a gelation kinetics in minutes time scale. Remaining experimental challenges for this system are a finer modulation of gelation kinetics and streamlining the synthesis of the prepolymer. Here we present the possibility to tune the gelation kinetics by introducing an electron-withdrawing substituent at p-position of the aryl MS ring. This variant also presents synthetic advantages. We study the influence of the p-substituent on the physicochemical properties of MS/SH crosslinked hydrogels, and their performance for cell encapsulation. We compare these properties with the PEG-MS variant containing an electron-donating linker. The new star poly(ethylene glycol)-4-(5-(methylsulfonyl)-1H-tetrazol-1-yl)benzamide (PEG4-CONH-TzMS) shows superior properties as cell encapsulating hydrogel in terms of ease of mixing polymer precursors, faster gelation, homogenous cell distribution and high enzymatic stability.
Junker, Fabian | Rupf, Stephan | Schindler, Paula Marie | Wilden, Cedric | Hohl, Mathias | Ruiz-Gómez, Gloria | Pisabarro, M. Teresa | Wrublewsky, Selina | Bickelmann, Caroline | Berhorst, Charlotte | Alansary, Dalia | Wieland, Ben | Bischoff, Markus | Lee, Poh Soo | Moeller, Stephanie | Berg, Albrecht | Dancker, Tobias A. | Lauterbach, Marcel A. | Ganse, Bergita | Prates Roma, Leticia | Steudter, Therese | Metzger, Wolfgang | Tschernig, Thomas | Ampofo, Emmanuel | Laschke, Matthias W. | Hannig, Matthias | Rother, Sandra
DOI:
Excessive protease activity and impaired tissue regeneration are hallmarks of many disease states. Elevated matrix metalloproteinase-9 (MMP-9) plays a key role in adverse tissue remodeling by excessively degrading extracellular matrix (ECM) components and growth factors. Tissue inhibitor of metalloproteinase-3 (TIMP-3) regulates ECM turnover, and its bioavailability is influenced by glycosaminoglycans (GAGs). This study aimed to develop a methacrylated gelatin (GelMA)-based hydrogel functionalized with acrylated sulfated hyaluronan (sHAc) as a TIMP-3 delivery system to decrease ECM degradation under pathophysiological conditions. sHAc incorporation enhanced hydrogel stiffness, reduced degradation rates and yielded sustained TIMP-3 release for up to 28 days. Molecular modeling and surface plasmon resonance demonstrated preferential binding of TIMP-3 to sHAc over hyaluronan methacrylates, together providing a molecular rationale for the reduced and sustained release of TIMP-3 from sHAc-containing hydrogels. Angiogenesis-related functional assays, supported by molecular modeling studies, indicate that sHAc modulates the anti-angiogenic activity of TIMP-3 by altering vascular endothelial growth factor receptor-associated signaling, while preserving metalloproteinase inhibition. Released TIMP-3 from GelMA/sHAc hydrogels retained bioactivity, effectively inhibiting MMP-9 activity and mitigating ECM degradation in-vitro and in human ex-vivo models. In a murine subcutaneous implantation model, sHAc-functionalized TIMP-3-loaded hydrogels were associated with reduced inflammatory cell presence and altered vascular- and matrix-related tissue signatures compared with GelMA controls. These findings underscore the potential of sHAc-functionalized GelMA hydrogels as biomaterials for therapeutics delivery, offering controlled TIMP-3 release and sustained bioactivity to promote ECM stability and on-demand MMP inhibition. This system represents a promising strategy for addressing the challenges of excessive MMP activity.
Muhovic, Berina | Puertas Bartolomé, María | Teruel Enrico, Lara Luana | Dupont, Louise | Jonas, Alain M. | Glinel, Karine | Del Campo, Aránzazu | Wittmann, Christoph
DOI:
Living therapeutic materials (LTMs) are an emerging class of biomaterials that integrate living cells within engineered polymer matrices to provide dynamic and responsive functionalities. In this study, we engineered the robust, nonpathogenic, and GRAS-certified microorganism Corynebacterium glutamicum into a multifunctional biofactory for LTM applications. Using synthetic biology, we designed and constructed C. glutamicum strains capable of sensing, reporting, and producing the extremolyte ectoine. Ectoine is a clinically used compatible solute with cytoprotective and anti-inflammatory properties that is widely applied in dermatological formulations, nasal sprays, and ophthalmic preparations for the treatment of inflammatory and stress-related conditions. The engineered strains were further encapsulated in polymer-based living materials, including membrane-in-gel patches and core–shell hydrogel systems, to create skin-compatible and ocular-applicable therapeutic platforms. We developed genetic biosensors that detect diaminobutyric acid (DABA), a key intermediate in the ectoine biosynthesis pathway, to enable the time-resolved monitoring of cellular function. These biosensors, which are integrated with fluorescence and enzymatic reporter systems, allowed the noninvasive visualization of metabolic activity. Encapsulation strategies were optimized to ensure high metabolic activity, structural stability, and biocontainment, along with the controlled release of ectoine for potential applications in drug delivery and protective therapies. This work highlights the potential of C. glutamicum as a versatile platform for next-generation LTMs, offering precise monitoring and targeted therapeutic capabilities toward multifunctional living materials for precision medicine and environmental biosensing applications.



