Publikationen
Shim, Hwirim | Budak, Özil | Haug, V. | Widmaier, M. | Presser, Volker
DOI:
We explore different electrolytes containing lithium perchlorate (LiClO4) and lithium bis(trifluoromethylsulfonyl)imide (LiTFSI) salts in various solvents at different temperatures to associate the electrochemical stability of lithium titanium oxide (LTO)/lithium manganese oxide (LMO)-activated carbon (AC) composite Li-ion hybrid supercapacitors. By varying the solvents and operation temperature, we were able to detect a correlation between the electrochemical performance and the conductivity of each electrolyte. To test the electrochemical stability and longevity of the electrolytes in such a system, full-cells were analyzed via a voltage floating for 10 h at charged state (2.8 V) of the full-cell. Energy and power performance were the highest for acetonitrile (ACN) containing electrolytes due to their high lithium-ion conductivity. Additionally, the longevity of LiClO4 in 3-methoxypropionitrile (3-MPN) was superior to all tested electrolytes at ambient temperature with 97% capacity retention and energy efficiency of about 86% after 500 h of voltage floating. LiClO4 in a mixture of ACN and ethylene carbonate (EC) was the most stable electrolyte at high temperatures with 70% capacity retention after 500 h voltage floating at 60°C and 50% capacity retention after 250 h voltage floating. The longevity of LiTFSI in ACN and LiClO4 in 3-MPN electrolyte demonstrated a capacity retention of 97% and 92% after 500 h voltage floating, respectively, at low temperature (−10 °C).
Srimuk, Pattarachai | Wang, Lei | Budak, Öznil | Presser, Volker
DOI:
Electrochemical processes enable a new generation of energy-efficient desalination technologies. While ion electrosorption via capacitive deionization is only suitable for brackish water with low molar strength, the use of Faradaic materials capable of reversible ion intercalation or conversion reactions allows energy-efficient removal of ions from seawater. However, the limited charge transfer/storage capacity of Faradaic materials indicates an upper limit for their desalination applications. Therefore, a new electrochemical concept must be explored to exceed the current state-of-the-art results and to push the desalination capacity beyond 100–200 mgNaCl/gelectrode. In this proof-of-concept work, we introduce the new concept of using metal–air battery technology for desalination. We do so by presenting performance data for zinc–air desalination (ZAD) in 600 mM NaCl. The ZAD cell provides a desalination capacity of 0.9–1.0 mgNaCl/cm2 (normalized to the membrane area; corresponding to 1300 mgNaCl/gZn) with a charge efficiency of 70% when charging/discharging the cell at 1 mA/cm2. The energy consumption of ZAD is 68–92 kJ/mol.
Talande, Smita V. | Bakandritsos, Aristides | Zdražil, Lukáš | Jakubec, Petr | Mohammadi, Elmira | Tomanec, Ondřej | Otyepka, Michal | Presser, Volker | Zbořil, Radek | Tuček, Jiří
DOI:
To meet the future demands for off-grid power, high-performance electrochemical energy storage based on earth-abundant materials is essential. Supercapacitors are attractive in this sense due to their sustainable carbon-based architecture, rapid charging/discharging, and long cycle-life in comparison to battery chemistries. However, hybridizing carbon electrodes with inorganic phases is intensively explored in supercapacitor research to mitigate their low energy content. Iron sulfides are attractive because they are non-toxic and composed of earth-abundant elements, but, despite their hydrophobic nature, they have only been studied in aqueous electrolytes, limiting the energy content due to the narrow voltage stability window of water. Here, exploiting a rapid growth method and a highly functionalized graphene support, we strongly immobilized greigite (Fe3S4) nanoparticles with an ultrasmall size which could not be attained in the absence of graphene. The respective supercapacitor cell was found significantly more electroactive in the ionic liquid electrolyte than in water, boosting the energy content. Furthermore, greigite has high conductivity and fast surface faradaic reactions due to the enzyme-mimicking triple redox state of its thiocubane basic structural unit. Thus, fully reversible and fast redox processes in the expanded voltage-window of the ionic liquid also endowed excellent rate capability, cycling stability, and power. The work demonstrates a pathway, not previously explored, whereby greigite/graphene hybrids can surpass in these aspects top-rated supercapacitor materials.
Torkamanzadeh, Mohammad | Wang, Lei | Zhang, Yuan | Budak, Öznil | Srimuk, Pattarachai | Presser, Volker
DOI:
Two-dimensional, layered transition metal carbides (MXenes) are an intriguing class of intercalation-type electrodes for electrochemical applications. The ability for preferred counterion uptake qualifies MXenes as an attractive material for electrochemical desalination. Our work explores Ti3C2Tx-MXene paired with activated carbon in such a way that both electrodes operate in an optimized potential range. This is accomplished by electrode mass balancing and control over the cell voltage. Thereby, we enable effective remediation of saline media with low (brackish) and high (seawater-like) ionic strength by using 20 and 600 mM aqueous NaCl solutions. It is shown that MXene/activated-carbon asymmetric cell design capitalizes on the permselective behavior of MXene in sodium removal, which in turn forces carbon to mirror the same behavior in the removal of chloride ions. This has minimized the notorious co-ion desorption of carbon in highly saline media (600 mM NaCl) and boosted the charge efficiency from 4% in a symmetric activated-carbon/activated-carbon cell to 85% in a membrane-less asymmetric MXene/activated-carbon cell. Stable electrochemical performance for up to 100 cycles is demonstrated, yielding average desalination capacities of 8 and 12 mg/g, respectively, for membrane-less MXene/activated-carbon cells in NaCl solutions of 600 mM (seawater-level) and 20 mM (brackish-water-level). In the case of the 20 mM NaCl solutions, surprising charge efficiency values of over 100% have been obtained, which is attributed to the role of MXene interlayer surface charges.
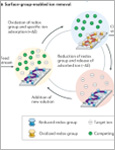
Srimuk, Pattarachai | Su, Xiao | Yoon, Jeyong | Aurbach, Doron | Presser, Volker
DOI:
Reversible electrochemical processes are a promising technology for energy-efficient water treatment. Electrochemical desalination is based on the compensation of electric charge by ionic species, through which the ions are immobilized and, thereby, removed from a feed-water stream flowing through a desalination cell. For decades, electrochemical desalination has focused on the use of carbon electrodes, but their salt-removal ability is limited by the mechanism of ion electrosorption at low molar concentrations and low charge-storage capacity. Recently, charge-transfer materials, often found in batteries, have demonstrated much larger charge-storage capacities and energy-efficient desalination at both low and high molar strengths. In this Review, we assess electrochemical-desalination mechanisms and materials, including ion electrosorption and charge-transfer processes, namely, ion binding with redox-active polymers, ion insertion, conversion reactions and redox-active electrolytes. Furthermore, we discuss performance metrics and cell architectures, which we decouple from the nature of the electrode material and the underlying mechanism to show the versatility of cell-design concepts. These charge-transfer processes enable a wealth of environmental applications, ranging from potable-water generation and industrial-water remediation to lithium recovery and heavy-metal-ion removal.
Zhang, Yuan | Srimuk, Pattarachai | Aslan, Mesut | Gallei, Markus | Presser, Volker
DOI:
In this work, carbon electrode modified polymer membrane and free-standing polymer membranes are applied for desalination via capacitive deionization with microporous activated carbon. The desalination performance under different concentrations (20 mM and 600 mM) of feed water and different voltage operation mode (zero-volt and voltage-reversal operation). For the ion exchange membrane coating under 20 mM NaCl solution at a cell voltage of 0/+1 V, the desalination capacity reached 22.7 mg/g at the charge efficiency of 142.1%. In the case of the free-standing membrane, under 600 mM NaCl solution at a voltage range of −1/+1 V, the desalination capacity reached 26.0 mg/g with a charge efficiency of 61.5%. These results not only show the possibility of membrane capacitive deionization (MCDI) application in seawater desalination but also reveals different ion transport mechanisms of the coated membrane and free-standing membranes under different molar strength of feed water.
Fleischmann, Simon | Dörr, Tobias S. | Frank, Anna | Hieke, Stefan W. | Doblas-Jimenez, David | Scheu, Christina | Oliveira, Peter William de | Kraus, Tobias | Presser, Volker
DOI:
Abstract Transition metal dichalcogenides are attractive two-dimensional electrode materials for electrochemical energy storage devices due to their high reversible charge storage capacity. Hybridization of these materials with carbon promises enhanced performance by facilitating the access to reactive sites and extended mechanical stabilization. Herein, we introduce a NbS2/C hybrid material exhibiting a gyroidal microstructure synthesized through macromolecular co-assembly of a tailored block copolymer and an organometallic niobium precursor and subsequent sulfidation. Our synthesis allows the preparation of mechanically stable monoliths with NbS2 nanocrystals engulfed in a highly porous carbon shell. Due to the curvature of the gyroidal structure, abundant reactive sites are exposed that lead to an attractive performance in a lithium-containing electrolyte with a capacity of up to 400 mAh/g.
Abbas, Qamar | Gollas, Bernhard | Presser, Volker
DOI:
The Faradaic processes related to electrochemical water reduction at the nanoporous carbon electrode under negative polarization are reduced when the concentration of aqueous sodium nitrate (NaNO3) is increased or the temperature is decreased. This effect enhances the relative contribution of ion electrosorption to the total charge storage process. Hydrogen chemisorption is reduced in aqueous 8.0 m NaNO3 due to the low degree of hydration of the Na+ cation; consequently, less free water is available for redox contributions, driving the system to exhibit electrical double-layer capacitive characteristics. Hydrogen adsorption/desorption is facilitated in 1.0 m NaNO3 due to the high molar ratio. The excess of water shifts the local pH in carbon nanopores to neutral values, giving rise to a high overpotential for dihydrogen evolution in the latter. The dilution effect on local pH shift in 1.0 m NaNO3 can be reduced by decreasing the temperature. A symmetric activated carbon cell assembled with 8.0 m NaNO3 exhibits a high capacitance and coulombic efficiency, a larger contribution of ion electrosorption to the overall charge storage process, and a stable capacitance performance at 1.6 V.
Budak, Öznil | Srimuk, Pattarachai | Tolosa, Aura | Fleischmann, Simon | Lee, Juhan | Hieke, Stefan W. | Frank, Anna | Scheu, Christina | Presser, Volker
DOI:
Abstract We present a facile two-step synthesis of vanadium (III) oxide/carbon core/shell hybrid material for application as lithium-ion battery electrode. The first step is a thermal treatment of a mixture of vanadium carbide (VC) and NiCl2 ⋅ 6H2O at 700 °C in an inert gas atmosphere. Elemental nickel obtained from decomposing NiCl2 ⋅ 6H2O served as a catalyst to trigger the local formation of graphitic carbon. In a second step, residual nickel was removed by washing the material in aqueous HCl. By replacing NiCl2 ⋅ 6H2O with anhydrous NiCl2, we obtained a hybrid material of vanadium carbide-derived carbon and a vanadium carbide core. Material characterization revealed a needle-like morphology of the rhombohedral V2O3 along with two carbon species with a different degree of graphitic ordering. We varied the NiCl2 ⋅ 6H2O-to-VC ratio, and the optimized material yielded a capacity of 110 mAh ⋅ g−1 at 2.5 A ⋅ g−1 which increased to 225 mAh ⋅ g−1 at 0.1 A ⋅ g−1 after 500 cycles in the potential range of 0.01-3.00 V vs. Li/Li+. This enhanced performance is in stark contrast to the loss of lithium uptake capacity when using commercially available V2O3 mixed with carbon black, where 93 % of the initial capacity was lost after 50 cycles.
Fleischmann, Simon | Pfeifer, Kristina | Widmaier, Mathias | Shim, Hwirim | Budak, Öznil | Presser, Volker
DOI:
Negative electrode materials that possess fast lithium insertion kinetics are in high demand for high power lithium-ion batteries and hybrid supercapacitor applications. In this work, hydrogen titanium oxides are synthesized by a proton exchange reaction with sodium titanium oxide, resulting in the H2Ti3O7 phase. We show that a gradual water release in four steps yields intermediate phases of hydrogen titanate with different degrees of interlayer protonation. In addition, a synthesis route using zinc nitrate is explored yielding H2Ti3O7 with a high rutile content. This material dehydrates already at a lower temperature, resulting in a lamellar rutile titania phase. The hydrogen titanate materials with partially protonated interlayers are tested as negative electrodes in a lithium-ion battery and hybrid supercapacitor setup, showing an improved performance compared to the fully protonated phases. The performance in half-cells reaches around 168 mAh/g, with high retention of 42 mAh/g at 10 A/g. This translates to an energy of 88 Wh/kg for a full-cell with a maximum power of 9.2 kW/kg and high cycling stability over 1000 cycles.

