Unsere Inspiration ist die Anpassungsfähigkeit von Organismen und den Materialien, aus denen sie aufgebaut sind, an wechselnde Umweltbedingungen. Pflanzen passen ihr Wachstum an die Lichtverhältnisse an, Bakterien entwickeln Resistenzen gegen Antibiotika oder Knochen werden durch Belastung stärker. Grundlage für diese Anpassungsfähigkeit ist eine faszinierende Signalverarbeitung der Organismen: Durch molekulare Sensoren werden Umweltbedingungen wahrgenommen, die Signale werden prozessiert und mit dem genetischen Programm des Organismus integriert, um am Ende eine passgenaue Reaktion auszulösen.
In unserer Forschung verwenden wir diese molekularen informationsverarbeitenden Mechanismen, um die Funktion und Eigenschaften von Zellen und Materialien gezielt zu steuern. Dies eröffnet neuartige Möglichkeiten in der grundladen- und anwendungsorientierten Forschung.
Mehr Informationen finden Sie auf unserer englischsprachigen Seite.

Mitarbeiter/innen










Publikationen

Schneider, N. | Wieland, F. G. | Kong, D. | Fischer, A. A. M. | Hörner, M. | Timmer, J. | Ye, H. | Weber, Wilfried
DOI:
Light-inducible gene switches represent a key strategy for the precise manipulation of cellular events in fundamental and applied research. However, the performance of widely used gene switches is limited due to low tissue penetrance and possible phototoxicity of the light stimulus. To overcome these limitations, we engineer optogenetic synthetic transcription factors to undergo liquid-liquid phase separation in close spatial proximity to promoters. Phase separation of constitutive and optogenetic synthetic transcription factors was achieved by incorporation of intrinsically disordered regions. Supported by a quantitative mathematical model, we demonstrate that engineered transcription factor droplets form at target promoters and increase gene expression up to fivefold. This increase in performance was observed in multiple mammalian cells lines as well as in mice following in situ transfection. The results of this work suggest that the introduction of intrinsically disordered domains is a simple yet effective means to boost synthetic transcription factor activity. Copyright © 2021 The Authors, some rights reserved.
Hörner, M. | Jerez-Longres, C. | Hudek, A. | Hook, S. | Yousefi, O. S. | Schamel, W. W. A. | Hörner, C. | Zurbriggen, M. D. | Ye, H. | Wagner, H. J. | Weber, Wilfried
DOI:
Methodologies for the controlled delivery of genetic information into target cells are of utmost importance for genetic engineering in both fundamental and applied research. However, available methods for efficient gene transfer into user-selected or even single cells suffer from low throughput, the need for complicated equipment, high invasiveness, or side effects by off-target viral uptake. Here, we engineer an adeno-associated viral (AAV) vector system that transfers genetic information into native target cells upon illumination with cell-compatible red light. This OptoAAV system allows adjustable and spatially resolved gene transfer down to single-cell resolution and is compatible with different cell lines and primary cells. Moreover, the sequential application of multiple OptoAAVs enables spatially resolved transduction with different transgenes. The approach presented is likely extendable to other classes of viral vectors and is expected to foster advances in basic and applied genetic research. Copyright © 2021 The Authors, some rights reserved.
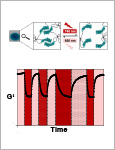
Hörner, M. | Raute, K. | Hummel, B. | Madl, J. | Creusen, G. | Thomas, O. S. | Christen, E. H. | Hotz, N. | Gübeli, R. J. | Engesser, R. | Rebmann, B. | Lauer, J. | Rolauffs, B. | Timmer, J. | Schamel, W. W. A. | Pruszak, J. | Römer, W. | Zurbriggen, M. D. | Friedrich, C. | Walther, A. | Minguet, S. | Sawarkar, R. | Weber, Wilfried
DOI:
Interrogation and control of cellular fate and function using optogenetics is providing revolutionary insights into biology. Optogenetic control of cells is achieved by coupling genetically encoded photoreceptors to cellular effectors and enables unprecedented spatiotemporal control of signaling processes. Here, a fast and reversibly switchable photoreceptor is used to tune the mechanical properties of polymer materials in a fully reversible, wavelength-specific, and dose- and space-controlled manner. By integrating engineered cyanobacterial phytochrome 1 into a poly(ethylene glycol) matrix, hydrogel materials responsive to light in the cell-compatible red/far-red spectrum are synthesized. These materials are applied to study in human mesenchymal stem cells how different mechanosignaling pathways respond to changing mechanical environments and to control the migration of primary immune cells in 3D. This optogenetics-inspired matrix allows fundamental questions of how cells react to dynamic mechanical environments to be addressed. Further, remote control of such matrices can create new opportunities for tissue engineering or provide a basis for optically stimulated drug depots. © 2019 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim

Ates, H. C. | Mohsenin, H. | Wenzel, C. | Glatz, R. T. | Wagner, H. J. | Bruch, R. | Hoefflin, N. | Spassov, S. | Streicher, L. | Lozano-Zahonero, S. | Flamm, B. | Trittler, R. | Hug, M. J. | Köhn, M. | Schmidt, J. | Schumann, S. | Urban, G. A. | Weber, Wilfried | Dincer, C.
DOI:
Personalized antibiotherapy ensures that the antibiotic concentration remains in the optimal therapeutic window to maximize efficacy, minimize side effects, and avoid the emergence of drug resistance due to insufficient dosing. However, such individualized schemes need frequent sampling to tailor the blood antibiotic concentrations. To optimally integrate therapeutic drug monitoring (TDM) into the clinical workflow, antibiotic levels can either be measured in blood using point-of-care testing (POCT), or can rely on noninvasive sampling. Here, a versatile biosensor with an antibody-free assay for on-site TDM is presented. The platform is evaluated with an animal study, where antibiotic concentrations are quantified in different matrices including whole blood, plasma, urine, saliva, and exhaled breath condensate (EBC). The clearance and the temporal evaluation of antibiotic levels in EBC and plasma are demonstrated. Influence of matrix effects on measured drug concentrations is determined by comparing the plasma levels with those in noninvasive samples. The system's potential for blood-based POCT is further illustrated by tracking ß‑lactam concentrations in untreated blood samples. Finally, multiplexing capabilities are explored successfully for multianalyte/sample analysis. By enabling a rapid, low-cost, sample-independent, and multiplexed on-site TDM, this system can shift the paradigm of “one‑size-fits-all” strategy. © 2021 The Authors. Advanced Materials published by Wiley-VCH GmbH
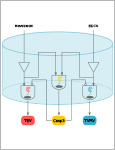
Mohsenin, Hasti | Wagner, Hanna J. | Rosenblatt, Marcus | Kemmer, Svenja | Dreppe, Friedel | Huesgen, Pitter | Timmer, Jens | Weber, Wilfried
DOI:
Synthetic biology applies concepts from electrical engineering and information processing to endow cells with computational functionality. Transferring the underlying molecular components into materials and wiring them according to topologies inspired by electronic circuit boards has yielded materials systems that perform selected computational operations. However, the limited functionality of available building blocks is restricting the implementation of advanced information-processing circuits into materials. Here, a set of protease-based biohybrid modules the bioactivity of which can either be induced or inhibited is engineered. Guided by a quantitative mathematical model and following a design-build-test-learn (DBTL) cycle, the modules are wired according to circuit topologies inspired by electronic signal decoders, a fundamental motif in information processing. A 2-input/4-output binary decoder for the detection of two small molecules in a material framework that can perform regulated outputs in form of distinct protease activities is designed. The here demonstrated smart material system is strongly modular and can be used for biomolecular information processing for example in advanced biosensing or drug delivery applications.


