We study and orchestrate how synthetic materials interact with living cells. We engineer cell-instructive environments and material-based solutions for zero-waste therapeutic solutions
Our group develops hydrogel materials with programmed and tunable properties designed to encapsulate and instruct living cells. We study how living cells and inert matter interact and how these interactions can be exploited to direct cellular functions and ultimately result in therapeutic advantages. We cooperate with synthetic biologists, biophysicists, drug developers and clinicians to explore the application potential of our developments, with a focus on new materials for ophthalmic drug delivery. We contribute to INM’s competence fields opto-interactive and bio-intelligent materials. Our research addresses biomedical needs.

Contact
Team Members






Research
Hydrogels with latent properties
By integrating phototriggers and light-responsive molecular motors in polymeric networks, we develop 4D hydrogels with optoregulated (bio)chemical activity, crosslinking, degradation, or mechanoactuation. These are used for biophysical studies of cell response to changes in the biochemical and mechanical signals of the extracellular environment.
Model Cellular Microenvironments
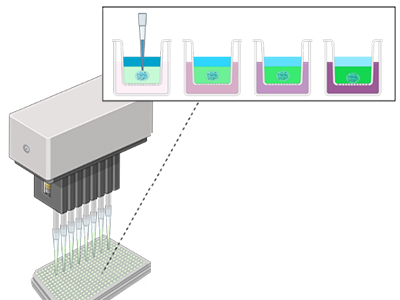
We build synthetic models of cell-matrix and cell-cell interfaces with encoded biochemical, mechanical and dimensional signals. To accelerate discovery, we develop material microarrays for high-throughput biophysical experimentation and apply them to study multifactorial cell responses.
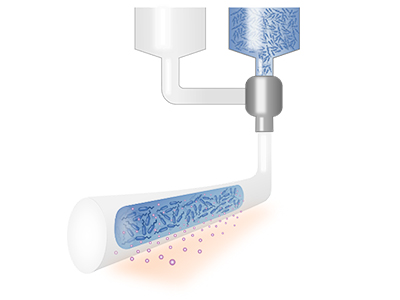
Living Therapeutic Devices
We develop bioinks and apply biofabrication technologies for functional and safe encapsulation of cells in medical devices. We focus on self-replenishable living therapeutic materials which integrate drug biofactories and have unlimited therapeutic release. We aim for innovation in ocular therapeutics with living, self-replenishable drug-eluting contact lenses.
Partner

Selected Publications
Sankaran, Shrikrishnan | Becker, Judith | Wittmann, Christoph | del Campo, Aránzazu
DOI:
Abstract On-demand and long-term delivery of drugs are common requirements in many therapeutic applications, not easy to be solved with available smart polymers for drug encapsulation. This work presents a fundamentally different concept to address such scenarios using a self-replenishing and optogenetically controlled living material. It consists of a hydrogel containing an active endotoxin-free Escherichia coli strain. The bacteria are metabolically and optogenetically engineered to secrete the antimicrobial and antitumoral drug deoxyviolacein in a light-regulated manner. The permeable hydrogel matrix sustains a viable and functional bacterial population and permits diffusion and delivery of the synthesized drug to the surrounding medium at quantities regulated by light dose. Using a focused light beam, the site for synthesis and delivery of the drug can be freely defined. The living material is shown to maintain considerable levels of drug production and release for at least 42 days. These results prove the potential and flexibility that living materials containing engineered bacteria can offer for advanced therapeutic applications.
Sankaran, Shrikrishnan | del Campo, Aránzazu
DOI:
Abstract Developing materials to encapsulate and deliver functional proteins inside the body is a challenging yet rewarding task for therapeutic purposes. High production costs, mostly associated with the purification process, short-term stability in vivo, and controlled and prolonged release are major hurdles for the clinical application of protein-based biopharmaceuticals. In an attempt to overcome these hurdles, herein, the possibility of incorporating bacteria as protein factories into a material and externally controlling protein release using optogenetics is demonstrated. By engineering bacteria to express and secrete a red fluorescent protein in response to low doses of blue light irradiation and embedding them in agarose hydrogels, living materials are fabricated capable of releasing proteins into the surrounding medium when exposed to light. These bacterial hydrogels allow spatially confined protein expression and dosed protein release over several weeks, regulated by the area and extent of light exposure. The possibility of incorporating such complex functions in a material using relatively simple material and genetic engineering strategies highlights the immense potential and versatility offered by living materials for protein-based biopharmaceutical delivery.
Farrukh, Aleeza | Paez, Julieta I. | del Campo, Aránzazu
DOI:
Abstract A strategy for spatial and temporal regulation of ligand presentation within a biomaterial, and the consequent site- and time-specific cellular responses in 4D cell cultures are presented. The key molecular component is a light-activatable adhesive peptidomimetic (cyclo Arg-Gly-Asp-phe-Cys, RGDfC) modified with the two-photon photocleavable group (p-methoxynitrobiphenyl, PMNB) used to functionalize a hydrogel. A scanning laser at 740 nm defines the 4D presentation of active RGD ligands within the gel, and directs basic cellular processes of embedded cells in situ. The excellent photochemical properties of the PMNB photoremovable group allows direct photomanipulation of the cellular environment without appreciable damage of the embedded cells. Light-directed migration of fibroblasts within a crosslinked poly(ethylene glycol) (PEG) hydrogel, and sequential, light-regulated angiogenesis with human umbilical vein endothelial cells (HUVECs) in 4D constructs is demonstrated. The materials presented here represent unique microenvironments to reconstruct dynamic changes in the composition of the extracellular space of cells that occur in in vivo tissues.
Dohle, Eva | Singh, Smriti | Nishigushi, Akihiro | Fischer, Thorsten | Wessling, Matthias | Möller, Martin | Sader, Robert | Kasper, Jennifer Y. | Ghanaati, Shahram | Kirkpatrick, C. James
DOI:
The development of an in vitro model resembling the alveolar-capillary barrier might be a highly beneficial tool to study lung physiology as well as the immune response of the lung to infection or after exposure to nanoparticles. This study is based on an in vitro alveolar barrier developed on a basement membrane mimic, composed of ultrathin nanofiber meshes generated via electrospinning using bioresorbable poly(ɛ-caprolactone). As cellular components, NCI H441, resembling the alveolar epithelial cells, and ISO-HAS-1, an endothelial cell line, were used to perform bipolar coculture experiments for a total cultivation period of 14 days. In addition to immunohistochemical and immunofluorescent studies, transepithelial electrical resistance (TER) and transport capabilities of the in vitro model system were investigated. Alveolar barrier function could be clearly determined for the postulated bipolar coculture system on the basement membrane mimic, since TER increased during the course of bipolar cultivation. Furthermore, to gain first insights into possible lung inflammatory reactions in vitro, this coculture model was further expanded by a human leukemia monocyte cell line (THP-1). This triple-culture system was able to maintain adequately the barrier properties of the bipolar coculture, thus making this in vitro model consisting of epithelial, endothelial, and immune cells on a basement membrane mimic a promising basis for further studies in tissue engineering.
Farrukh, Aleeza | Fan, Wenqiang | Zhao, Shifang | Salierno, Marcelo | Paez, Julieta I. | Del Campo, Aranzazu
DOI:
Abstract Neuro-regeneration after trauma requires growth and reconnection of neurons to reestablish information flow in particular directions across the damaged tissue. To support this process, biomaterials for nerve tissue regeneration need to provide spatial information to adhesion receptors on the cell membrane and to provide directionality to growing neurites. Here, photoactivatable adhesive peptides based on the CASIKVAVSADR laminin peptidomimetic are presented and applied to spatiotemporal control of neuronal growth to biomaterials in vitro. The introduction of a photoremovable group [6-nitroveratryl (NVOC), 3-(4,5-dimethoxy-2-nitrophenyl)butan-2-yl (DMNPB), or 2,2′-((3′-(1-hydroxypropan-2-yl)-4′-nitro-[1,1′-biphenyl]-4-yl)azanediyl)bis(ethan-1-ol) (HANBP)] at the amino terminal group of the K residue temporally inhibited the activity of the peptide. The bioactivity was regained through controlled light exposure. When used in neuronal culture substrates, the peptides allowed light-based control of the attachment and differentiation of neuronal cells. Site-selective irradiation activated adhesion and differentiation cues and guided seeded neurons to grow in predefined patterns. This is the first demonstration of ligand-based light-controlled interaction between neuronal cells and biomaterials.
Farrukh, Aleeza | Zhao, Shifang | del Campo, Aránzazu
DOI:
Strategies for neural tissue repair heavily depend on our ability to temporally reconstruct the natural cellular microenvironment of neural cells. Biomaterials play a fundamental role in this context, as they provide the mechanical support for cells to attach and migrate to the injury site, as well as fundamental signals for differentiation. This review describes how different cellular processes (attachment, proliferation, and (directional) migration and differentiation) have been supported by different material parameters, in vitro and in vivo. Although incipient guidelines for biomaterial design become visible, literature in the field remains rather phenomenological. As in other fields of tissue regeneration, progress will depend on more systematic studies on cell-materials response, better understanding on how cells behave and understand signals in their natural milieu from neurobiology studies, and the translation of this knowledge into engineered microenvironments for clinical use.
Farrukh, Aleeza | Zhao, Shifang | Paez, Julieta I. | Kavyanifar, Atria | Salierno, Marcelo | Cavalié, Adolfo | del Campo, Aránzazu
DOI:
The ability to guide the growth of neurites is relevant for reconstructing neural networks and for nerve tissue regeneration. Here, a biofunctional hydrogel that allows light-based directional control of axon growth in situ is presented. The gel is covalently modified with a photoactivatable derivative of the short laminin peptidomimetic IKVAV. This adhesive peptide contains the photoremovable group 2-(4′-amino-4-nitro-[1,1′-biphenyl]-3-yl)propan-1-ol (HANBP) on the Lys rest that inhibits its activity. The modified peptide is highly soluble in water and can be simply conjugated to −COOH containing hydrogels via its terminal −NH2 group. Light exposure allows presentation of the IKVAV adhesive motif on a soft hydrogel at desired concentration and at defined position and time point. The photoactivated gel supports neurite outgrowth in embryonic neural progenitor cells culture and allows site-selective guidance of neurites extension. In situ exposure of cell cultures using a scanning laser allows outgrowth of neurites in desired pathways.
Nair, Roshna V. | Farrukh, Aleeza | del Campo, Aránzazu
DOI:
Abstract The integrin α5β1 is overexpressed in colon, breast, ovarian, lung and brain tumours, and has been identified as key component in mechanosensing. In order to study how dynamic changes in α5β1 engagement affect cellular behaviour, photoactivatable derivatives of α5β1-specific ligands are presented in this article. A photoremovable protecting group (PRPG) was introduced into the ligand structure at a relevant position for integrin recognition. The presence of the chromophore temporarily inhibited ligand bioactivity. Light exposure at a cell-compatible dose efficiently cleaved the protecting group and restored functionality. The photoactive ligand had an azide end-functional group for covalent immobilisation onto biomaterials by click chemistry. Selective cell responses (attachment, spreading, migration) to the activated ligand on the surface are achieved by controlled exposure to light, at similar levels to the native ligand. Spatial and temporal control of the cellular response is demonstrated, including the possibility of in situ activation. Photoactivatable integrin-selective ligands in model microenvironments will allow the study of cellular behaviour in response to changes in the activation of individual integrins as consequence of dynamic variations in matrix composition.
Rohles, Christina Maria | Gläser, Lars | Kohlstedt, Michael | Gießelmann, Gideon | Pearson, Samuel | del Campo, Aránzazu | Becker, Judith | Wittmann, Christoph
DOI:
In the present work, we established the bio-based production of glutarate, a carbon-5 dicarboxylic acid with recognized value for commercial plastics and other applications, using metabolically engineered Corynebacterium glutamicum. The mutant C. glutamicum AVA-2 served as a starting point for strain development, because it secreted small amounts of glutarate as a consequence of its engineered 5-aminovalerate pathway. Starting from AVA-2, we overexpressed 5-aminovalerate transaminase (gabT) and glutarate semialdehyde dehydrogenase (gabD) under the control of the constitutive tuf promoter to convert 5-aminovalerate further to glutarate. The created strain GTA-1 formed glutarate as a major product, but still secreted 5-aminovalerate as well. This bottleneck was tackled at the level of 5-aminovalerate re-import. The advanced strain GTA-4 overexpressed the newly discovered 5-aminovalerate importer NCgl0464 and formed glutarate from glucose in a yield of 0.27 mol mol−1. In a fed-batch process, GTA-4 produced more than 90 g L−1 glutarate from glucose and molasses based sugars in a yield of up to 0.70 mol mol−1 and a maximum productivity of 1.8 g L−1 h−1, while 5-aminovalerate was no longer secreted. The bio-based glutaric acid was purified to >99.9% purity. Interfacial polymerization and melt polymerization with hexamethylenediamine yielded bionylon-6,5, a polyamide with a unique structu
Sankaran, Shrikrishnan | Zhao, Shifang | Muth, Christina | Paez, Julieta | del Campo, Aránzazu
DOI:
Abstract Living materials are an emergent material class, infused with the productive, adaptive, and regenerative properties of living organisms. Property regulation in living materials requires encoding responsive units in the living components to allow external manipulation of their function. Here, an optoregulated Escherichia coli (E. coli)-based living biomaterial that can be externally addressed using light to interact with mammalian cells is demonstrated. This is achieved by using a photoactivatable inducer of gene expression and bacterial surface display technology to present an integrin-specific miniprotein on the outer membrane of an endotoxin-free E. coli strain. Hydrogel surfaces functionalized with the bacteria can expose cell adhesive molecules upon in situ light-activation, and trigger cell adhesion. Surface immobilized bacteria are able to deliver a fluorescent protein to the mammalian cells with which they are interacting, indicating the potential of such a bacterial material to deliver molecules to cells in a targeted manner.



