We study and orchestrate how synthetic materials interact with living cells. We engineer cell-instructive environments and material-based solutions for zero-waste therapeutic solutions
Our group develops hydrogel materials with programmed and tunable properties designed to encapsulate and instruct living cells. We study how living cells and inert matter interact and how these interactions can be exploited to direct cellular functions and ultimately result in therapeutic advantages. We cooperate with synthetic biologists, biophysicists, drug developers and clinicians to explore the application potential of our developments, with a focus on new materials for ophthalmic drug delivery. We contribute to INM’s competence fields opto-interactive and bio-intelligent materials. Our research addresses biomedical needs.

Contact
Team Members






Research
Hydrogels with latent properties
By integrating phototriggers and light-responsive molecular motors in polymeric networks, we develop 4D hydrogels with optoregulated (bio)chemical activity, crosslinking, degradation, or mechanoactuation. These are used for biophysical studies of cell response to changes in the biochemical and mechanical signals of the extracellular environment.
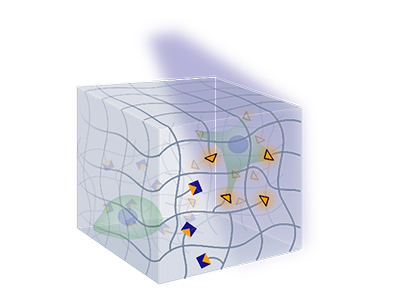
Model Cellular Microenvironments
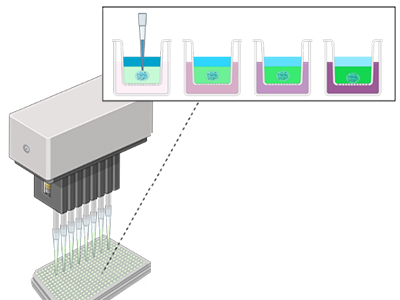
We build synthetic models of cell-matrix and cell-cell interfaces with encoded biochemical, mechanical and dimensional signals. To accelerate discovery, we develop material microarrays for high-throughput biophysical experimentation and apply them to study multifactorial cell responses.
Living Therapeutic Devices
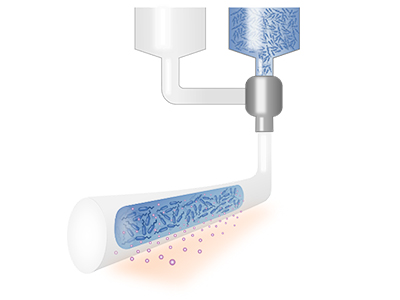
We develop bioinks and apply biofabrication technologies for functional and safe encapsulation of cells in medical devices. We focus on self-replenishable living therapeutic materials which integrate drug biofactories and have unlimited therapeutic release. We aim for innovation in ocular therapeutics with living, self-replenishable drug-eluting contact lenses.
Partner

Selected Publications
Wlodarczyk-Biegun, Malgorzata | Zielinski, Piotr | Gladysz, Magdalena | Hofman, Anno | Wu, Xixi | Villiou, Maria | Koch, Marcus | del Campo, Aránzazu | Kamperman, Marleen
DOI:
DOI:Włodarczyk-Biegun | Villiou, Maria | Koch, Klaus Peter | Muth, Christina | Wang | Ott | del Campo
DOI:
The permeability of the human trabecular meshwork (HTM) regulates eye pressure via a porosity gradient across its thickness modulated by stacked layers of matrix fibrils and cells. Changes in HTM porosity are associated with increases in intraocular pressure and the progress of diseases such as glaucoma. Engineered HTMs could help to understand the structure–function relation in natural tissues and lead to new regenerative solutions. Here, melt electrowriting (MEW) is explored as a biofabrication technique to produce fibrillar, porous scaffolds that mimic the multilayer, gradient structure of native HTM. Poly(caprolactone) constructs with a height of 125–500 μm and fiber diameters of 10–12 μm are printed. Scaffolds with a tensile modulus between 5.6 and 13 MPa and a static compression modulus in the range of 6–360 kPa are obtained by varying the scaffold design, that is, the density and orientation of the fibers and number of stacked layers. Primary HTM cells attach to the scaffolds, proliferate, and form a confluent layer within 8–14 days, depending on the scaffold design. High cell viability and cell morphology close to that in the native tissue are observed. The present work demonstrates the utility of MEW for reconstructing complex morphological features of natural tissues.
Alhayek, Alaa | Khan, Essak S. | Schönauer, Esther | Däinghaus, Tobias | Shafiei, Roya | Voos, Katrin | Han, Mitchell K. L | Ducho, Christian | Posselt, Gernot | Wessler, Silja | Brandstetter, Hans | Haupenthal, Jörg | del Campo, Aránzazu | Hirsch, Anna K. H.
DOI:
Abstract Despite the progress in surgical techniques and antibiotic prophylaxis, opportunistic wound infections with Bacillus cereus remain a public health problem. Secreted toxins are one of the main factors contributing to B. cereus pathogenicity. A promising strategy to treat such infections is to target these toxins and not the bacteria. Although the exoenzymes produced by B. cereus are thoroughly investigated, little is known about the role of B. cereus collagenases in wound infections. In this report, the collagenolytic activity of secreted collagenases (Col) is characterized in the B. cereus culture supernatant (csn) and its isolated recombinantly produced ColQ1 is characterized. The data reveals that ColQ1 causes damage on dermal collagen (COL). This results in gaps in the tissue, which might facilitate the spread of bacteria. The importance of B. cereus collagenases is also demonstrated in disease promotion using two inhibitors. Compound 2 shows high efficacy in peptidolytic, gelatinolytic, and COL degradation assays. It also preserves the fibrillar COLs in skin tissue challenged with ColQ1, as well as the viability of skin cells treated with B. cereus csn. A Galleria mellonella model highlights the significance of collagenase inhibition in vivo.
Feng, Jun | Zheng, Yijun | Jiang, Qiyang | Włodarczyk-Biegun, Małgorzata K. | Pearson, Samuel | del Campo, Aránzazu
DOI:
Abstract Advances in optogenetics and the increasing use of implantable devices for therapies and health monitoring are driving demand for compliant, biocompatible optical waveguides and scalable methods for their manufacture. Molding, thermal drawing, and dip-coating are the most prevalent approaches in recent literature. Here the authors demonstrate that extrusion printing at room temperature can be used for continuous fabrication of compliant optical waveguides with polydimethylsiloxane (PDMS) core and crosslinked Pluronic F127-diacrylate (Pluronic-DA) cladding. The optical fibers are printed from fluid precursor inks and stabilized by physical interactions and photoinitiated crosslinking in the Pluronic-DA. The printed fibers show optical loss values of 0.13–0.34 dB cm–1 in air and tissue within the wavelength range of 405–520 nm. The fibers have a Young's Modulus (Pluronic cladding) of 150 kPa and can be stretched to more than 5 times their length. The optical loss of the fibers shows little variation with extension. This work demonstrates how printing can simplify the fabrication of compliant and stretchable devices from materials approved for clinical use. These can be of interest for optogenetic or photopharmacology applications in extensible tissues, like muscles or heart.
Jin, Minye | Gläser, Alisa | Paez, Julieta I.
DOI:
Stimuli-responsive hydrogels are smart materials that respond to variations caused by external stimuli and that are currently exploited for biomedical applications such as biosensing, drug delivery and tissue engineering. The development of stimuli-responsive hydrogels with defined user control is relevant to realize materials with advanced properties. Recently, our group reported firefly luciferin-inspired hydrogel matrices for 3D cell culture. This platform exhibited advantages like rapid gelation rate and tunability of mechanical and biological properties. However, this first molecular design did not allow fine control of the gelation onset, which restricts application as a cell-encapsulating matrice with injectable and processable properties. In this article, we endow the firefly luciferin-inspired hydrogels with redox-triggering capability, to overcome the limitations of the previous system and to widen its application range. We achieve this goal by introducing protected macromers as hydrogel polymeric precursors that can be activated in the presence of a mild reductant, to trigger gel formation in situ with a high degree of control. We demonstrate that the regulation of molecular parameters (e.g., structure of the protecting group, reductant type) and environmental parameters (e.g., pH, temperature) of the deprotection reaction can be exploited to modulate materials properties. This redox-triggerable system enables precise control over gelation onset and kinetics, thus facilitating its utilization as an injectable hydrogel without negatively impacting its cytocompatibility. Our findings expand the current toolkit of chemically-based stimuli-responsive hydrogels.
Jin, Minye | Koçer, Gülistan | Paez, Julieta I.
DOI:
There is an increasing interest in coupling reactions for cross-linking of cell-encapsulating hydrogels under biocompatible, chemoselective, and tunable conditions. Inspired by the biosynthesis of luciferins in fireflies, here we exploit the cyanobenzothiazole-cysteine (CBT-Cys) click ligation to develop polyethylene glycol hydrogels as tunable scaffolds for cell encapsulation. Taking advantage of the chemoselectivity and versatility of CBT-Cys ligation, a highly flexible gel platform is reported here. We demonstrate luciferin-inspired hydrogels with important advantages for cell encapsulation applications: (i) gel precursors derived from inexpensive reagents and with good stability in aqueous solution (>4 weeks), (ii) adjustable gel mechanics within physiological ranges (E = 180–6240 Pa), (iii) easy tunability of the gelation rate (seconds to minutes) by external means, (iv) high microscale homogeneity, (v) good cytocompatibility, and (iv) regulable biological properties. These flexible and robust CBT-Cys hydrogels are proved as supportive matrices for 3D culture of different cell types, namely, fibroblasts and human mesenchymal stem cells. Our findings expand the toolkit of click chemistries for the fabrication of tunable biomaterials.
Koçer, Gülistan | Albino, Inês M. C. | Verheijden, Mark L. | Jonkheijm, Pascal
DOI:
Endothelial cells play a central role in the vascular system, where their function is tightly regulated by both cell-extracellular matrix (e.g., via integrins) and cell–cell interactions (e.g., via cadherins). In this study, we incorporated cholesterol-modified integrin and N-cadherin peptide binding ligands in fluid supported lipid bilayers. Human umbilical vein endothelial cell adhesion, spreading and vinculin localization in these cells were dependent on ligand density. One composition led to observe a higher extent of cell spreading, where cells exhibited extensive lamellipodia formation and a qualitatively more distinct N-cadherin localization at the cell periphery, which is indicative of N-cadherin clustering and a mimic of cell–cell contact formation. The results can be used to reconstitute the endothelial-pericyte interface on biomedical devices and materials.
Yang, Wenjuan | Denger, Andreas | Diener, Caroline | Küppers, Frederic | Soriano-Baguet, Leticia | Schäfer, Gertrud | Yanamandra, Archana K. | Zhao, Renping | Knörck, Arne | Schwarz, Eva C. | Hart, Martin | Lammert, Frank | Roma, Leticia Prates | Brenner, Dirk | Christidis, Grigorios | Helms, Volkhard | Meese, Eckart | Hoth, Markus | Qu, Bin
DOI:
TNF-related apoptosis inducing ligand (TRAIL) is expressed on cytotoxic T lymphocytes (CTLs) and TRAIL is linked to progression of diabetes. However, the impact of high glucose on TRAIL expression and its related killing function in CTLs still remains largely elusive. Here, we report that TRAIL is substantially up-regulated in CTLs in environments with high glucose (HG) both in vitro and in vivo. Non-mitochondrial reactive oxygen species, NFκB and PI3K/Akt are essential in HG-induced TRAIL upregulation in CTLs. TRAIL<sup>high</sup> CTLs induce apoptosis of pancreatic beta cell line 1.4E7. Treatment with metformin and vitamin D reduces HG-enhanced expression of TRAIL in CTLs and coherently protects 1.4E7 cells from TRAIL-mediated apoptosis. Our work suggests that HG-induced TRAIL<sup>high</sup> CTLs might contribute to the destruction of pancreatic beta cells in a hyperglycemia condition.
Zhang, Jingnan | Zhao, Renping | Li, Bin | Farrukh, Aleeza | Hoth, Markus | Qu, Bin | del Campo, Aránzazu
DOI:
The analysis of T cell responses to mechanical properties of antigen presenting cells (APC) is experimentally challenging at T cell-APC interfaces. Soft hydrogels with adjustable mechanical properties and biofunctionalization are useful reductionist models to address this problem. Here, we report a methodology to fabricate micropatterned soft hydrogels with defined stiffness to form spatially confined T cell/hydrogel contact interfaces at micrometer scale. Using automatized microcontact printing we prepared arrays of anti-CD3 microdots on poly(acrylamide) hydrogels with Young's Modulus in the range of 2 to 50 kPa. We optimized the printing process to obtain anti-CD3 microdots with constant area (50 µm2, corresponding to 8 µm diameter) and comparable anti-CD3 density on hydrogels of different stiffness. The anti-CD3 arrays were recognized by T cells and restricted cell attachment to the printed areas. To test functionality of the hydrogel-T cell contact, we analyzed several key events downstream of T cell receptor (TCR) activation. Anti-CD3 arrays on hydrogels activated calcium influx, induced rearrangement of the actin cytoskeleton, and led to Zeta-chain-associated protein kinase 70 (ZAP70) phosphorylation. Interestingly, upon increase in the stiffness, ZAP70 phosphorylation was enhanced, whereas the rearrangements of F-actin (F-actin clearance) and phosphorylated ZAP70 (ZAP70/pY centralization) were unaffected. Our results show that micropatterned hydrogels allow tuning of stiffness and receptor presentation to analyze TCR mediated T cell activation as function of mechanical, biochemical, and geometrical parameters.
Rodrigo-Navarro, Aleixandre | Sankaran, Shrikrishnan | Dalby, Matthew J. | del Campo, Aránzazu | Salmeron-Sanchez, Manuel
DOI:
Biomaterials have evolved from inert materials that lack interaction with the body to biologically active, instructive materials that host and provide signals to surrounding cells and tissues. Engineered living materials contain living cells (responsive function) and polymeric matrices (scaffolding function) and, thus, can be designed as active and response biomaterials. In this Review, we discuss engineered living materials that incorporate microorganisms as the living, bioactive component. Microorganisms can provide complex responses to environmental stimuli, and they can be genetically engineered to allow user control over responses and integration of numerous inputs. The engineered microorganisms can either generate their own matrix, such as in biofilms, or they can be incorporated in matrices using various technologies, such as coating, 3D printing, spinning and microencapsulation. We highlight biomedical applications of such engineered living materials, including biosensing, wound healing, stem-cell-based tissue engineering and drug delivery, and provide an outlook to the challenges and future applications of engineered living materials.



