We study and orchestrate how synthetic materials interact with living cells. We engineer cell-instructive environments and material-based solutions for zero-waste therapeutic solutions
Our group develops hydrogel materials with programmed and tunable properties designed to encapsulate and instruct living cells. We study how living cells and inert matter interact and how these interactions can be exploited to direct cellular functions and ultimately result in therapeutic advantages. We cooperate with synthetic biologists, biophysicists, drug developers and clinicians to explore the application potential of our developments, with a focus on new materials for ophthalmic drug delivery. We contribute to INM’s competence fields opto-interactive and bio-intelligent materials. Our research addresses biomedical needs.

Contact
Team Members
Research
Hydrogels with latent properties
By integrating phototriggers and light-responsive molecular motors in polymeric networks, we develop 4D hydrogels with optoregulated (bio)chemical activity, crosslinking, degradation, or mechanoactuation. These are used for biophysical studies of cell response to changes in the biochemical and mechanical signals of the extracellular environment.
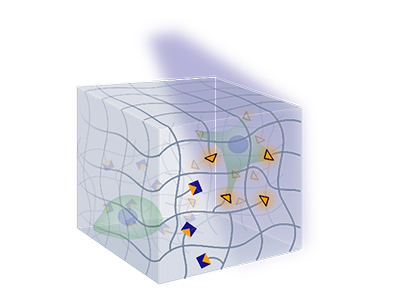
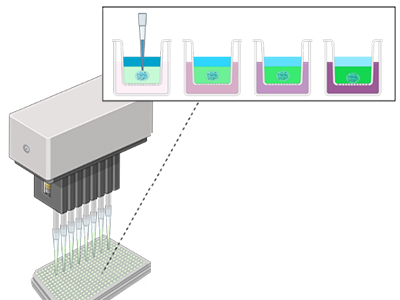
Model Cellular Microenvironments
We build synthetic models of cell-matrix and cell-cell interfaces with encoded biochemical, mechanical and dimensional signals. To accelerate discovery, we develop material microarrays for high-throughput biophysical experimentation and apply them to study multifactorial cell responses.
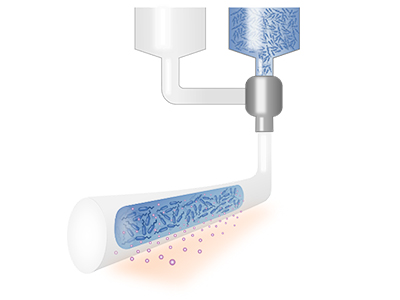
Living Therapeutic Devices
We develop bioinks and apply biofabrication technologies for functional and safe encapsulation of cells in medical devices. We focus on self-replenishable living therapeutic materials which integrate drug biofactories and have unlimited therapeutic release. We aim for innovation in ocular therapeutics with living, self-replenishable drug-eluting contact lenses.
Partner

Selected Publications
Kasper, Jennifer Y. | Laschke, Matthias W. | Koch, Marcus | Alibardi, Lorenzo | Magin, Thomas M. | Niessen, Carien M. | del Campo, Aránzazu
DOI:
The hierarchical design of the toe pad surface in geckos and its reversible adhesiveness have inspired material scientists for many years. Micro- and nano-patterned surfaces with impressive adhesive performance have been developed to mimic gecko's properties. While the adhesive performance achieved in some examples has surpassed living counterparts, the durability of the fabricated surfaces is limited and the capability to self-renew and restore function—inherent to biological systems—is unimaginable. Here the morphogenesis of gecko setae using skin samples from the Bibron´s gecko (Chondrodactylus bibronii) is studied. Gecko setae develop as specialized apical differentiation structures at a distinct cell–cell layer interface within the skin epidermis. A primary role for F-actin and microtubules as templating structural elements is necessary for the development of setae's hierarchical morphology, and a stabilization role of keratins and corneus beta proteins is identified. Setae grow from single cells in a bottom layer protruding into four neighboring cells in the upper layer. The resulting multicellular junction can play a role during shedding by facilitating fracture of the cell–cell interface and release of the high aspect ratio setae. The results contribute to the understanding of setae regeneration and may inspire future concepts to bioengineer self-renewable patterned adhesive surfaces.
Trujillo, Sara | Kasper, Jennifer Y. | Miguel-Jimenez, Adrián De | Abt, Britta | Bauer, Alina | Mekontso Ngaffo, Joelle A. | Pearson, Samuel | del Campo, Aránzazu
DOI:
Methylsulfone derivatized poly(ethylene) glycol (PEG) macromers can be biofunctionalized with thiolated ligands and cross-linked with thiol-based cross-linkers to obtain bioactive PEG hydrogels for in situ cell encapsulation. Methylsulfonyl-thiol (MS-SH) reactions present several advantages for this purpose when compared to other thiol-based cross-linking systems. They proceed with adequate and tunable kinetics for encapsulation, they reach a high conversion degree with good selectivity, and they generate stable reaction products. Our previous work demonstrated the cytocompatibility of cross-linked PEG-MS/thiol hydrogels in contact with fibroblasts. However, the cytocompatibility of the in situ MS-SH cross-linking reaction itself, which generates methylsulfinic acid as byproduct at the cross-linked site, remains to be evaluated. These studies are necessary to evaluate the potential of these systems for in vivo applications. Here we perform an extensive cytocompatibility study of PEG hydrogels during in situ cross-linking by the methylsulfonyl-thiol reaction. We compare these results with maleimide–thiol cross-linked PEGs which are well established for cell culture and in vivo experiments and do not involve the release of a byproduct. We show that fibroblasts and endothelial cells remain viable after in situ polymerization of methylsulfonyl-thiol gels on the top of the cell layers. Cell viability seems better than after in situ cross-linking hydrogels with maleimide–thiol chemistry. The endothelial cell proinflammatory phenotype is low and similar to the one obtained by the maleimide–thiol reaction. Finally, no activation of monocytes is observed. All in all, these results demonstrate that the methylsulfonyl-thiol chemistry is cytocompatible and does not trigger high pro-inflammatory responses in endothelial cells and monocytes. These results make methylsulfonyl-thiol chemistries eligible for in vivo testing and eventually clinical application in the future.
Bhusari, Shardul | Hoffmann, Maxi | Herbeck-Engel, Petra | Sankaran, Shrikrishnan | Wilhelm, Manfred | del Campo, Aránzazu
DOI:
Pluronic (Plu) hydrogels mixed with variable fractions of Pluronic diacrylate (PluDA) have become popular matrices to encapsulate bacteria and control their growth in engineered living materials. Here we study the rheological response of 30 wt.% Plu/PluDA hydrogels with PluDA fraction between 0 and 1. We quantify the range of viscoelastic properties that can be covered in this system by varying in the PluDA fraction. We present stress relaxation and creep-recovery experiments and describe the variation of the critical yield strain/stress, relaxation and recovery parameters of Plu/PluDA hydrogels as function of the covalent crosslinking degree using the Burgers and Weilbull models. The analyzed hydrogels present two stress relaxations with different timescales which can be tuned with the covalent crosslinking degree. We expect this study to help users of Plu/PluDA hydrogels to estimate the mechanical properties of their systems, and to correlate them with the behaviour of bacteria in future Plu/PluDA devices of similar composition.



