The Research Group Materials-Host Interactions investigates how Living Therapeutic Materials (LTMs) interact with in vitro tissue models. We focus on understanding the risks associated with LTMs, to facilitate their safe and sustainable translation.

Mitarbeiter/innen
Research
Investigation of Living Therapeutic Materials
in vitro for pre-clinical development
We develop experimental methods to investigate LTM interactions with host tissue models in vitro. We develop compatible culture conditions for bacterial-based LTMs together with tissue models. We investigate effects that relate to LTM biocompatibility and safety, so we can quantify the probability of LTMs to cause unwanted side effects to the host.


Development of cell and tissue models in health and disease
We apply 2D and 3D cell culture methods for the fabrication of functional models that can be used to investigate host responses to different stimuli. We use molecular biology techniques to characterize phenotypical and morphological changes so we can determine the potential effects of different treatments (materials or other therapeutics).
Our cellular models
To understand specific interactions between LTMs (or other therapeutics) and their target tissue in the host, we have established several in vitro models, and we continue to develop new ones to incorporate to our portfolio. We focus on angiogenesis, inflammation and biological barriers like the cornea.
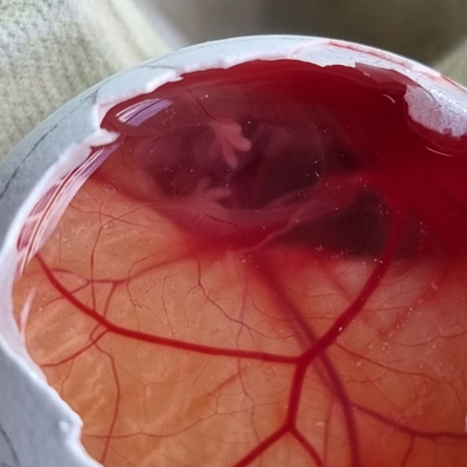
Investigation of angiogenic potential
We have developed 2D and 3D models to assess pro- and anti-angiogenic potential using primary human endothelial cells and dermal fibroblasts. We also use the chick chorioallantoic membrane assay, an ex vivo system for the assessment of new capillary formation. These models allow us to quantify several parameters during endothelial cell sprouting.


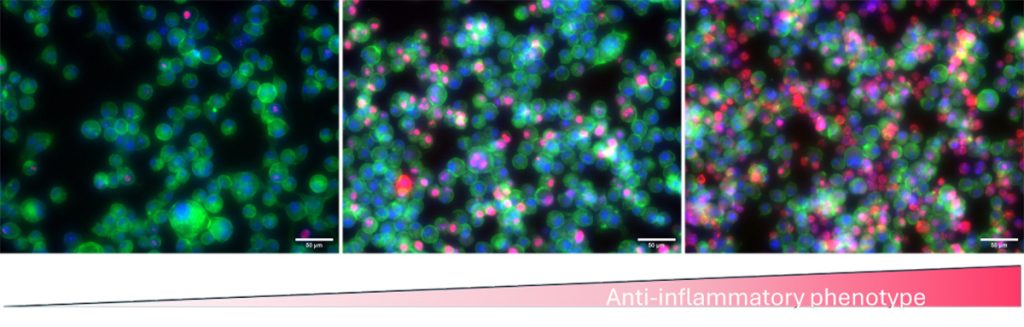
Models of endotoxemia
We are developing methods to assess inflammatory potential in vitro, by using cells involved in the foreign body reaction such as monocytes, macrophages and fibroblasts. We characterize phenotypic and morphological changes of these cells when in contact with LTMs or treated with different therapeutics. We use the monocyte activation test to detect the presence of endotoxin or non-endotoxin pyrogens, which can trigger fever in the host. We have developed an in vitro model of endotoxemia using macrophages, which can be used to investigate the mechanism of action of some therapeutics
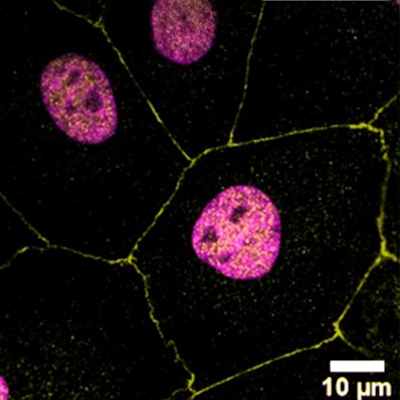
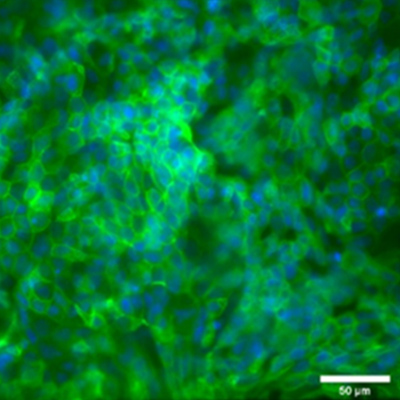
Biological barriers models – Cornea
We are developing different barrier models, with a focus on the cornea tissue. We use these models to investigate drug-delivery to the eye. The complexity of biological barriers (skin, gut, blood-brain barrier, eye) comes from their layered structure with different cells that are critical for the overall function of the tissue. Many biological barriers are in contact with the air and therefore complex 3D layered models at the air-liquid interface are leveraged to recapitulate these tissues. These models will help us understand for example, how LTMs can be applied to the eye.
Publikationen
Tadimarri, Varun S. | Blanch-Asensio, Marc | Deshpande, Ketaki | Baumann, Jonas | Baumann, Carole | Müller, Rolf | Trujillo, Sara | Sankaran, Shrikrishnan
DOI:
Engineered living materials (ELMs) made of bacteria in hydrogels have shown considerable promise for therapeutic applications through controlled and sustained release of complex biopharmaceuticals at low costs and with reduced wastage. While most therapeutic ELMs use E. coli due to its large genetic toolbox, most live biotherapeutic bacteria in development are lactic acid bacteria due to native health benefits they offer. Among these, lactobacilli form the largest family of probiotics with therapeutic potential in almost all sites of the body with a microbiome. A major factor limiting the use of lactobacilli in ELMs is their limited genetic toolbox. This study expands on recent work to expand the genetic programmability of probiotic Lactiplantibacillus plantarum WCFS1 for protein secretion and encapsulate it in a simple, cost-effective, and biocompatible core–shell alginate bead to develop an ELM. The controlled release of recombinant proteins is demonstrated, even up to 14 days from this ELM, thereby terming it PEARL – Protein Eluting Alginate with Recombinant Lactobacilli. Notably, lactobacillus encapsulation offered benefits like bacterial containment, protein release profile stabilization, and metabolite-induced cytotoxicity prevention. These findings demonstrate the mutual benefits of combining recombinant lactobacilli with alginate for the controlled and sustained release of proteins.
Desai, Krupansh | Garcia-Sanchez, Lorely | Amini, Maryam | Teruel Enrico, Lara Luana | Siegrist, Silke | Del Campo, Aránzazu | Trujillo, Sara
DOI:
In ophthalmology, living biomaterials have appeared as promising drug delivery and biosensor devices to tackle dynamic sensing and delivery of compounds. Their living character complicates their assessment with the also dynamic ocular tissues. The use of animal experiments increases complexity, and most animal ocular models are anatomically different from humans. Thus, in vitro ocular systems applied specifically to living biomaterials are required to assess their safety, compatibility and efficacy. Here, we report on an in vitro cornea model for co-cultures with Corynebacterium glutamicum-polyvinyl alcohol living biomaterials, which are reported as suitable living contact lenses, to study their cytocompatibility to the eye. We co-cultured this living biomaterial with human primary corneal cells (epithelial and fibroblasts) for 7 days, mimicking contact lens extended wear. We studied bacterial proliferation, biocontainment and biosafety. We investigated potential cytotoxicity and pro-inflammatory responses of living biomaterials to corneal cells. Our results revealed that the living biomaterial does not trigger cytotoxicity or pro-inflammatory phenotypes on corneal cells during the 7-day co-culture. We placed the living biomaterial on top of the corneal epithelium, observing no cytotoxic effects. Overall, these findings highlight the potential of in vitro investigations for living biomaterials and the applicability of these devices for ophthalmology purposes.
Munoz-Núnez, Carolina | Barco-Martín, A. | Deshpande, Ketaki | Schmidt, D. S. | Gonzalez-Garcia, Lola | Trujillo, Sara | Munoz-Bonilla, Alexander | Fernández-García, Marta
DOI:
In this study novel polymeric materials based on chitosan (CS) were synthesized by chemically modifying CS with two bioactive moieties: eugenol and a compound containing a thiazolium group. These modifications aimed to impart antioxidant and antimicrobial properties to the matrix. Additionally, the scaffolds were reinforced with chitin nanowhiskers (Nw) to improve their mechanical strength and stability. Porous three-dimensional scaffolds were fabricated via the freeze-drying process, resulting in highly interconnected pore networks suitable for cell infiltration and nutrient transport. Biological characterization revealed that the incorporation of the two bioactive groups significantly enhanced the antioxidant activity and antimicrobial efficacy against both Gram-positive and Gram-negative bacteria to the scaffolds. Mechanical testing demonstrated that the Nw reinforcement increased scaffold stiffness and resilience without compromising porosity. In vitro biological assays using fibroblasts showed favorable cytocompatibility and promoted sustained cell proliferation over three weeks. Fluorescence microscopy confirmed fibroblast adhesion and morphological adaptation within the scaffold architecture. Additionally, the scaffolds were evaluated for their immunomodulatory effects using macrophage cultures, revealing a balanced immune response with reduced proinflammatory signaling, which is critical for successful integration and reduced fibrosis in vivo. These results indicate that those are promising candidates for tissue engineering and regenerative medicine applications.
Mekontso Ngaffo, Joelle A. | Farrukh, Usama | Trujillo, Sara | Del Campo, Aránzazu
DOI:
Living Therapeutic Materials (LTMs) are a promising alternative to polymeric drug carriers for long term release of biotherapeutics. LTMs contain living drug biofactories that produce the drug using energy sources from the body fluids. To clarify their application potential, it is fundamental to adapt biocompatibility and cytotoxicity assays applied from non-living biomaterials and therapeutics to evaluate how LTMs interact with host cells. Here, we have established a first step in this direction, by developing a practical workflow to parallelize in vitro assessment of minimal safety and cytocompatibility properties of bacterial LTMs. It allows systematic monitoring and quantification of the dynamic evolution of the bacterial population (growth, metabolic activity) in parallel to quantify the response of different mammalian cells to LTM supernatants with regards to cytotoxicity and release of pro-inflammatory cytokines over a period of 7 days using a maximum of 10 samples. The protocol was tested with a Pluronic-based thin film containing ClearColi. The results show no cytotoxic effects of ClearColi containing hydrogels in three mammalian cell lines, and no induction of pro-inflammatory cytokines under the tested conditions. This workflow represents a first step in establishing a roadmap for the safety assessment of LTMs, and investigation of biocompatibility potential of future living therapeutic devices.
Desai, Krupansh K. | Sankaran, Shrikrishnan | Del Campo, Aránzazu | Trujillo, Sara
DOI:
Engineered living materials (ELMs), which usually comprise bacteria, fungi, or animal cells entrapped in polymeric matrices, offer limitless possibilities in fields like drug delivery or biosensing. Determining the conditions that sustain ELM performance while ensuring compatibility with ELM hosts is essential before testing them in vivo. This is critical to reduce animal experimentation and can be achieved through in vitro investigations. Currently, there are no standards that ensure ELM compatibility with host tissues. Towards this goal, we designed a 96-well plate-based screening method to streamline ELM growth across culture conditions and determine their compatibility potential in vitro. We showed proliferation of three bacterial species encapsulated in hydrogels over time and screened six different cell culture media. We fabricated ELMs in bilayer and monolayer formats and tracked bacterial leakage as a measure of ELM biocontainment. After screening, an appropriate medium was selected that sustained growth of an ELM, and it was used to study cytocompatibility in vitro. ELM cytotoxicity on murine fibroblasts and human monocytes was studied by adding ELM supernatants and measuring cell membrane integrity and live/dead staining, respectively, proving ELM cytocompatibility. Our work illustrates a simple setup to streamline the screening of compatible environmental conditions of ELMs with the host.
Desai, Krupansh K. | Mekontso, Joelle | Deshpande, Ketaki | Trujillo, Sara
DOI:
Advances in the past decades in materials science, biofabrication methods, and synthetic biology have given rise to new fields like living materials. A living material is a class of biohybrid composite with living elements, including bacteria, yeasts, fungi, and mammalian cells, integrated with nonliving components. (1−6) These materials combine the advantages of both living and nonliving components to generate novel functions such as responses to environmental parameters and syntheses of complex biomolecules. (7) The nonliving aspect combines diverse chemistries and manufacturing techniques to support or enhance the functions of the living part. (6) Living materials as therapeutics (Living Therapeutic Materials, LTMs) bring revolutionary options to diagnostic and therapeutic practice, offering a solution to life-concerning issues by life itself (Figure 1). Living Therapeutic Materials are revolutionizing classical drug delivery devices, as they can produce therapeutics long-term, in situ, and on demand. This represents a more sustainable way for treatment. To realize Living Therapeutic Materials in the clinic, more preclinical studies need to be carried out so the concerns in terms of safety are well understood and their capacity as a more efficient delivery system is assessed. In the past decade, there has been a rise in the number of proof-of-concept LTMs and yet, the preclinical investigation of these materials is just starting.
Deshpande, Ketaki | Tadimarri, Varun S. | Ramirez-Rangel, Juliette | Sankaran, Shrikrishnan | Trujillo, Sara
DOI:
Living therapeutics are attractive candidates to tackle the limitations of classically delivered therapeutic peptides, which are often poorly stable and require cost-intensive modifications. Their functional assessment is limited to animal experiments, which increase the complexity to evaluate the dynamic nature of these systems. Therefore, we developed an in vitro model of endotoxemia using macrophages to assess early-stage anti-inflammatory Living therapeutics. We refined the model based on three anti-inflammatory peptides (KCF-18, I6P7, and α-MSH) and identified suitable therapeutic concentrations and treatment durations. We applied the model to Lactiplantibacillus plantarum TF103, a probiotic engineered to secrete these peptides. The model revealed that Living therapeutics enhanced the effects of the peptides, requiring lower amounts of anti-inflammatory effects. This points to potential synergistic effects between peptides and bacteria. The model presented here allows the investigation of dynamic regimes, which could be useful in the development of complex systems such as the ones encountered in Living therapeutics.
DOI:
Increasing aging population, digital screen use, environmental factors, and sleep disorders have contributed to a rise in ophthalmic diseases. This has soared the demand for better ocular models that are more predictive and can be used to identify new pharmacological targets. Traditional models fail to recapitulate organ-level functionalities and present anatomical differences with human structures, therefore, organ-on-chip systems have emerged to tackle these limitations. Microfluidic devices is engineered to provide the layered structure that the ocular tissues require. This is combined with tight regulation of diffusion gradients and perfusion systems for toxicological analysis and drug screening applications. Incorporation of several cellular layers, motion to mimic blinking, or incorporation of ocular organoids in microfluidic devices are some of the advancements that the field has made. This work reviews the evolution of ocular microphysiological systems and discusses some challenges that could be undertaken by the organ-on-chip community.
Deshpande, Ketaki | Tadimarri, Varun S. | Trujillo, Sara
DOI:
The most common characteristic observed in numerous diseases like rheumatoid arthritis or psoriasis is chronic inflammation. Endotoxemia is an important factor in these conditions as it is triggered by prolonged exposure to lipopolysaccharide (LPS), leading to inflammation and immune dysregulation. Therapeutic peptides are promising options to treat these chronic diseases with inflammatory characteristics. However, the applicability of therapeutic peptides is limited due to their poor stability in the body, which is typically overcome by cost-intensive modifications. Living therapeutics are emerging as a more cost-effective strategy to tackle this limitation by engineering microbes to produce and deliver the peptides right where they are needed. We developed an in-vitro endotoxemia (and psoriatic) model to test living therapeutics secreting anti-inflammatory peptides: KCF-18, I6P7, α-MSH (secreted from a genetically modified lactic acid-free strain of Lactiplantibacillus plantarum (TF103)) on murine macrophages, characterized the dose-response effects of these peptides and performed multi-array cytokine analysis. The model revealed that this living therapeutic approach enhanced the effects of the peptides, requiring lower amounts to achieve anti-inflammatory effects. Notably, α-MSH secreted by TF103 L. plantarum achieved significant pathway suppression, comparable to or exceeding that of synthetic controls, without inducing cytotoxicity. This points to potential synergistic effects between the peptides and the intrinsic anti-inflammatory properties of lactic acid bacteria. We will expand the applicability potential of these anti-inflammatory living therapeutic materials in an in vitro model of psoriasis.
Puertas-Bartolomé, María | Gutierrez-Urrutia, Izabook | Teruel-Enrico, Lara Luana | Dong, Nguyen Cao | Desai, Krupansh K. | Trujillo, Sara | Wittmann, Christoph | del Campo, Aránzazu
DOI:
The increasing prevalence of dry eye syndrome in aging and digital societies compromises long-term contact lens (CL) wear and forces users to regular eye drop instillation to alleviate discomfort. Here a novel approach with the potential to improve and extend the lubrication properties of CLs is presented. This is achieved by embedding lubricant-secreting biofactories within the CL material. The self-replenishable reservoirs autonomously produce and release hyaluronic acid (HA), a natural lubrication and wetting agent, long term. The hydrogel matrix regulates the growth of the biofactories and the HA production, and allows the diffusion of nutrients and HA for at least 3 weeks. The continuous release of HA sustainably reduces the friction coefficient of the CL surface. A self-lubricating CL prototype is presented, where the functional biofactories are contained in a functional ring at the lens periphery, outside of the vision area. The device is cytocompatible and fulfils physicochemical requirements of commercial CLs. The fabrication process is compatible with current manufacturing processes of CLs for vision correction. It is envisioned that the durable-by-design approach in living CL could enable long-term wear comfort for CL users and minimize the need for lubricating eye drops.

