The Research Group Materials-Host Interactions investigates how Living Therapeutic Materials (LTMs) interact with in vitro tissue models. We focus on understanding the risks associated with LTMs, to facilitate their safe and sustainable translation.

Mitarbeiter/innen


Research
Investigation of Living Therapeutic Materials
in vitro for pre-clinical development
We develop experimental methods to investigate LTM interactions with host tissue models in vitro. We develop compatible culture conditions for bacterial-based LTMs together with tissue models. We investigate effects that relate to LTM biocompatibility and safety, so we can quantify the probability of LTMs to cause unwanted side effects to the host.


Development of cell and tissue models in health and disease
We apply 2D and 3D cell culture methods for the fabrication of functional models that can be used to investigate host responses to different stimuli. We use molecular biology techniques to characterize phenotypical and morphological changes so we can determine the potential effects of different treatments (materials or other therapeutics).
Our cellular models
To understand specific interactions between LTMs (or other therapeutics) and their target tissue in the host, we have established several in vitro models, and we continue to develop new ones to incorporate to our portfolio. We focus on angiogenesis, inflammation and biological barriers like the cornea.
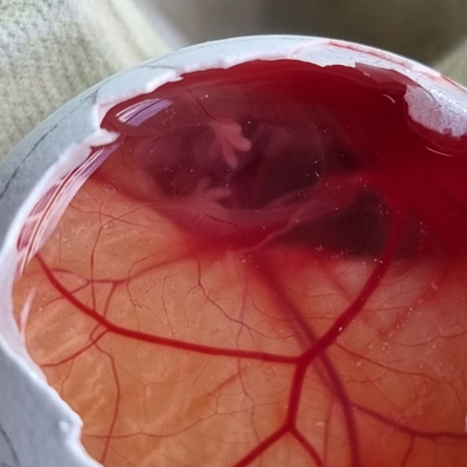
Investigation of angiogenic potential
We have developed 2D and 3D models to assess pro- and anti-angiogenic potential using primary human endothelial cells and dermal fibroblasts. We also use the chick chorioallantoic membrane assay, an ex vivo system for the assessment of new capillary formation. These models allow us to quantify several parameters during endothelial cell sprouting.


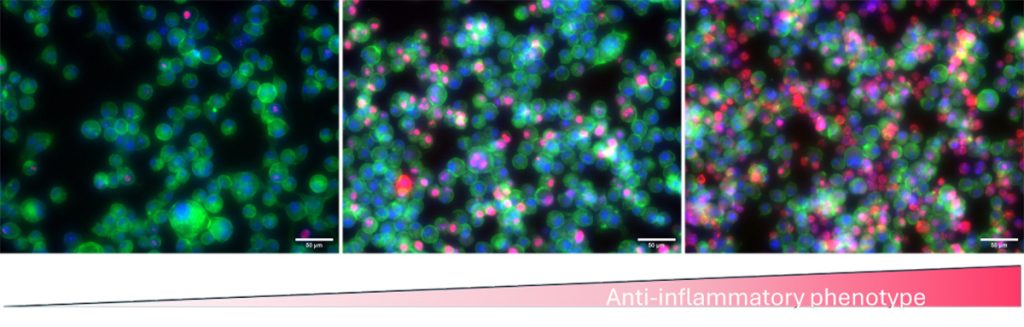
Models of endotoxemia
We are developing methods to assess inflammatory potential in vitro, by using cells involved in the foreign body reaction such as monocytes, macrophages and fibroblasts. We characterize phenotypic and morphological changes of these cells when in contact with LTMs or treated with different therapeutics. We use the monocyte activation test to detect the presence of endotoxin or non-endotoxin pyrogens, which can trigger fever in the host. We have developed an in vitro model of endotoxemia using macrophages, which can be used to investigate the mechanism of action of some therapeutics
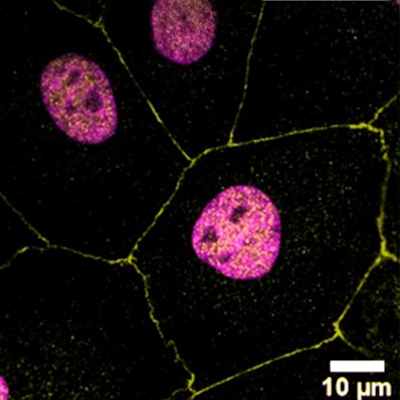
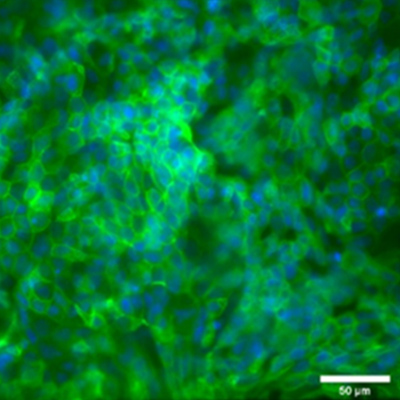
Biological barriers models – Cornea
We are developing different barrier models, with a focus on the cornea tissue. We use these models to investigate drug-delivery to the eye. The complexity of biological barriers (skin, gut, blood-brain barrier, eye) comes from their layered structure with different cells that are critical for the overall function of the tissue. Many biological barriers are in contact with the air and therefore complex 3D layered models at the air-liquid interface are leveraged to recapitulate these tissues. These models will help us understand for example, how LTMs can be applied to the eye.
Publikationen
Trujillo, Sara | Kasper, Jennifer Y. | Miguel-Jimenez, Adrián De | Abt, Britta | Bauer, Alina | Mekontso Ngaffo, Joelle A. | Pearson, Samuel | del Campo, Aránzazu
DOI:
Methylsulfone derivatized poly(ethylene) glycol (PEG) macromers can be biofunctionalized with thiolated ligands and cross-linked with thiol-based cross-linkers to obtain bioactive PEG hydrogels for in situ cell encapsulation. Methylsulfonyl-thiol (MS-SH) reactions present several advantages for this purpose when compared to other thiol-based cross-linking systems. They proceed with adequate and tunable kinetics for encapsulation, they reach a high conversion degree with good selectivity, and they generate stable reaction products. Our previous work demonstrated the cytocompatibility of cross-linked PEG-MS/thiol hydrogels in contact with fibroblasts. However, the cytocompatibility of the in situ MS-SH cross-linking reaction itself, which generates methylsulfinic acid as byproduct at the cross-linked site, remains to be evaluated. These studies are necessary to evaluate the potential of these systems for in vivo applications. Here we perform an extensive cytocompatibility study of PEG hydrogels during in situ cross-linking by the methylsulfonyl-thiol reaction. We compare these results with maleimide–thiol cross-linked PEGs which are well established for cell culture and in vivo experiments and do not involve the release of a byproduct. We show that fibroblasts and endothelial cells remain viable after in situ polymerization of methylsulfonyl-thiol gels on the top of the cell layers. Cell viability seems better than after in situ cross-linking hydrogels with maleimide–thiol chemistry. The endothelial cell proinflammatory phenotype is low and similar to the one obtained by the maleimide–thiol reaction. Finally, no activation of monocytes is observed. All in all, these results demonstrate that the methylsulfonyl-thiol chemistry is cytocompatible and does not trigger high pro-inflammatory responses in endothelial cells and monocytes. These results make methylsulfonyl-thiol chemistries eligible for in vivo testing and eventually clinical application in the future.
Monferrer, Ezequiel | Dobre, Oana | Trujillo, Sara | González Oliva, Mariana Azevedo | Trubert-Paneli, Alexandre | Acevedo-León, Delia | Noguera, Rosa | Salmeron-Sanchez, Manuel
DOI:
The tumor microenvironment plays an important role in cancer development and the use of 3D in vitro systems that decouple different elements of this microenvironment is critical for the study of cancer progression. In neuroblastoma (NB), vitronectin (VN), an extracellular matrix protein, has been linked to poor prognosis and appears as a promising therapeutic target. Here, we developed hydrogels that incorporate VN into 3D polyethylene glycol (PEG) hydrogel networks to recapitulate the native NB microenvironment. The stiffness of the VN/PEG hydrogels was modulated to be comparable to the in vivo values reported for NB tissue samples. We used SK-N-BE (2) NB cells to demonstrate that PEGylated VN promotes cell adhesion as the native protein does. Furthermore, the PEGylation of VN allows its crosslinking into the hydrogel network, providing VN retention within the hydrogels that support viable cells in 3D. Confocal imaging and ELISA assays indicate that cells secrete VN also in the hydrogels and continue to reorganize their 3D environment. Overall, the 3D VN-based PEG hydrogels recapitulate the complexity of the native tumor extracellular matrix, showing that VN-cell interaction plays a key role in NB aggressiveness, and that VN could potentially be targeted in preclinical drug studies performed on the presented hydrogels.


