Curriculum Vitae
Education
2017 – 2020
PhD in Biology (summa cum laude), Heidelberg University
Max Planck Institute for Medical Research (Prof. Joachim P. Spatz)
2015 – 2017
M.Sc. Molecular Biotechnology, Heidelberg University
Max Planck Institute for Intelligent Systems, Stuttgart, Germany
2011 – 2015
B.Sc. Molecular Biotechnology, Heidelberg University
Dept. for Biophysical Chemistry, Heidelberg University, Germany
Academic Positions
Since 2022
Research Group Leader
Leibniz Institute for New Materials, Saarbrücken Germany
Since 2022
Adjunct Research Group Leader
Max Planck Bristol Center for Minimal Biology, Bristol, UK
2021 – 2022
Marie Skłodowska Curie Individual Postdoctoral Research Fellow
Kennedy Institute of Rheumatology, University of Oxford, UK
2020 – 2021
Postdoctoral Scientist
Max Planck Institute for Medical Research, Heidelberg, Germany
Awards, Fellowships and Recognitions
- Alois Lauer Research Prize for Medicine, Alois Lauer Foundation
- Member of Junge Akademie Mainz, Academy of Sciences and Literature Mainz
- Emmy Noether Research Group Leader, German Science Foundation
- Daimler and Benz Foundation, Postdoctoral Fellow
- Otto Hahn Medal, Max Planck Society
- Young Investigator Award, International Society of Extracellular Vesicles
- Karl-von-Frisch Award, German Life Science Organization (vBio)
- Individual Fellowship, Marie Skłodowska Curie Program, European Commission
- Walter Benjamin Postdoctoral Fellowship, German Science Foundation
- Add-on Postdoctoral Fellowship, Joachim Herz Foundation
- PhD-Fellowship, Max Planck School Matter to Life
- PhD-Fellowship, Hartmut Hoffmann-Berling Graduate School
- Exchange Fellowship, Elisabeth Meurer Foundation
- Study Fellowship, German National Merit Fundation (Studienstiftung des Deutschen Volkes)
Publications

Burgstaller, Anna | Nink, Tamara | Walter, Niklas | Lopez Lopez, Erick Angel | Chang, Shin-Fang | Staufer, Oskar
DOI:
Synthetic cells have emerged as a novel biomimetic approach for studying fundamental cellular functions and enabling new therapeutic interventions. However, the potential to program synthetic cells into self-organized 3D collectives to replicate the structure and function of tissues has remained largely untapped. Here, self-assembly properties are engineered into synthetic cells to form millimeter-sized 3D lymphatic bottom-up tissues (lymphBUTs) with mechanical adaptability, metabolic activity, and hierarchical microstructural organization. It is demonstrated that primary human immune cells spontaneously infiltrate and functionally integrate into these synthetic lymph nodes to form living tissue hybrids. Applying lymphBUTs, it is shown that structured 3D organization and mechanical support drives T cell activation and the application of lymphBUTs for ex vivo expansion of regulatory CD8+ T cells is demonstrated. The study highlights the functional integration of living and non-living matter, advancing synthetic cell engineering toward 3D tissue structures.
Zhou, Xiangda | Zhang, Sijia | Yang, Wenjuan | Gonder, Susanne | Sadjadi, Zeinab | Piernitzki, Nils | Moter, Alina | Sharma, Shulagna | Largeot, Anne | Küchler, Nadja | Kaschek, Lea | Schäfer, Gertrud | Schwarz, Eva C. | Eichler, Hermann | Ullrich, Evelyn | Rieger, Heiko | Staufer, Oskar | Paggetti, Jérome | Moussay, Etienne | Hoth, Markus | Qu, Bin
DOI:
Natural killer (NK) cells are critical components of the first-line immune defense, responsible for eliminating tumorigenic cells. NK cell-based adoptive immunotherapy has gained increasing attention; however, cryopreservation, a standard technique for NK cell storage, significantly impairs NK cell cytotoxicity, particularly in physiological 3D environments. Here, we demonstrate that short-term co-culture with effector T cells markedly enhances NK cell motility and killing functionality. Notably, a brief 1-day co-culture is sufficient to restore cryopreservation-impaired NK cell functionality in 3D environments. This enhancement requires direct contact between T cells and NK cells, which facilitates localized high concentrations of IL-2 at the cell contact sites. To develop a controled, donor-independent solution, we demonstrate that synthetic T cells with surface-bound IL-2 exhibit superior efficiency in revitalizing cryopreserved NK cells. These findings uncover a previously unrecognized role for physical contact-mediated local IL-2 signaling and provide an efficient, cost-effective, and tunable strategy to rescue NK cell functionality post-cryopreservation, paving the way for more scalable, potent, and clinically viable NK cell-based immunotherapies.
Leithner, Alexander | Staufer, Oskar | Mitra, Tanmay | Liberta, Falk | Valvo, Salvatore | Kutuzov, Mikhail | Dada, Hannah | Spaeth, Jacob | Zhou, Weijie | Schiele, Felix | Reindl, Sophia | Nar, Herbert | Hoerer, Stefan | Crames, Maureen | Comeau, Stephen | Young, David | Low, Sarah | Jenkins, Edward | Davis, Simon J. | Klenerman, David | Nixon, Andrew | Pefaur, Noah | Wyatt, David | Dushek, Omer | Kasturirangan, Srinath | Dustin, Michael L.
DOI:
Bispecific T cell engagers (TcEs) link T cell receptors to tumor-associated antigens on cancer cells, forming cytotoxic immunological synapses (IS). Close membrane-to-membrane contact (≤13 nm) has been proposed as a key mechanism of TcE function. To investigate this and identify potential additional mechanisms, we compared four immunoglobulin G1-based (IgG1) TcE Formats (A–D) targeting CD3ε and Her2, designed to create varying intermembrane distances (A < B < C < D). Small-angle X-ray scattering (SAXS) and modeling of the conformational states of isolated TcEs and TcE–antigen complexes predicted close contacts (≤13 nm) for Formats A and B and far contacts (≥18 nm) for Formats C and D. In supported lipid bilayer (SLB) model interfaces, Formats A and B recruited, whereas Formats C and D repelled, CD2–CD58 interactions. Formats A and B also excluded bulky Quantum dots more effectively. SAXS also revealed that TcE–antigen complexes formed by Formats A and C were less flexible than complexes formed by Formats B and D. Functional data with Her2-expressing tumor cells showed cytotoxicity, surface marker expression, and cytokine release following the order A > B = C > D. In a minimal system for IS formation on SLBs, TcE performance followed the trend A = B = C > D. Addition of close contact requiring CD58 costimulation revealed phospholipase C-γ activation matching cytotoxicity with A > B = C > D. Our findings suggest that when adhesion is equivalent, TcE potency is determined by two parameters: contact distance and flexibility. Both the close/far-contact formation axis and the low/ high flexibility axis significantly impact TcE potency, explaining the similar potency of Format B (close contact/high flexibility) and C (far contact/low flexibility). Copyright © 2025 the Author(s).
Burgstaller, Anna | Madureira, Sara | Staufer, Oskar
DOI:
Tissue functions rely on complex structural, biochemical, and biomechanical cues that guide cellular behavior and organization. Synthetic cells, a promising new class of biomaterials, hold significant potential for mimicking these tissue properties using simplified, nonliving building blocks. Advanced synthetic cell models have already shown utility in biotechnology and immunology, including applications in cancer targeting and antigen presentation. Recent bottom-up approaches have also enabled synthetic cells to assemble into 3D structures with controlled intercellular interactions, creating tissue-like architectures. Despite these advancements, challenges remain in replicating multicellular behaviors and dynamic mechanical environments. Here, we review recent advancements in synthetic cell-based tissue formation and introduce a three-pillar framework to streamline the development of synthetic tissues. This approach, focusing on synthetic extracellular matrix integration, synthetic cell self-organization, and adaptive biomechanics, could enable scalable synthetic tissues engineering for regenerative medicine and drug development.
Piernitzki, Nils | Gao, Ning | Gasparoni, Gilles | Krauß, Louisa M. | Schulze-Hentrisch, Julia | Dustin, Michael | Schrul, Bianca | Györffy, Balázs | Mann, Stephen | Staufer, Oskar
DOI:
Self-assembly is a fundamental property of living matter that drives the three-dimensional organization of cell collectives such as tissues and organs. Here, the co-assembly of synthetic and natural cells is leveraged to create hybrid living 3D cancer cultures. We screen a range of synthetic cell models for their ability to form augmented tumoroids with artificial but controllable micro-environments, and show that the balance of inter- and extracellular adhesion and synthetic cell surface tension are key material properties driving integrated co-assembly. We demonstrate that synthetic cells based on droplet-supported lipid bilayers can establish artificial tumor immune microenvironments (ART-TIMEs), mimicking immunogenic signals within tumoroids and eliminating the need to integrate complex living immune cells. Using the ART-TIME approach, we identify a AhR-ARNT-mediated co-signaling mechanism between PD-1 and CD2 as a driver in immune evasion of pancreatic ductal adenocarcinoma. Our study advances the field of hybrid organoid engineering, offers opportunities for the construction and modelling of artificial tumour environments, and marks a step towards the design of functional living/non-living cytomimetic materials.
Piernitzki, Nils | Staufer, Oskar
DOI:
Extracellular vesicles (EVs) are lipid-membrane-enclosed particles released from cells, playing a pivotal role in cellular communication, particularly within the immune system. The fundamental molecular mechanisms through which EVs offer unique functionality for immunotherapeutic benefits are identified and reviewed. The focus is on three essential features, all rooted in the EV lipid membrane: immune receptor–ligand interactions at the EV membrane interface, the shielding of immunogenic cargo within the EVs, and the fusion of EVs with target cell membranes for direct cargo delivery. From this, how these distinct EV attributes, from their initial description and analysis in immune communication, have led to the development of novel immunotherapeutic strategies is traced. This review delves into how these strategies are applied in various immunotherapies, such as cancer immunotherapy, autoimmune diseases, infections, vaccinations, and graft-versus-host diseases, to modulate communication among different cell types for immune regulation. It is concluded by reviewing clinical trials involving EVs in immunotherapy that have effectively harnessed EVs' unique molecular mechanisms in clinical settings. Research and standardization efforts to maximize the potential impact of EVs on immunotherapy are further suggested.
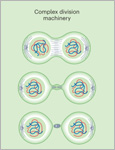
DOI:
In a major advancement for synthetic biology, dynamin A has been identified as a minimal component enabling cell division in synthetic cells, moving us one step nearer to realizing the ambition of creating synthetic life forms.
Jahnke, Kevin | Staufer, Oskar
DOI:
Extracellular vesicles (EVs), lipid-enclosed structures released by virtually all life forms, have gained significant attention due to their role in intercellular and interorganismal communication. Despite their recognized importance in disease processes and therapeutic applications, fundamental questions about their primary function remain. Here, we propose a different perspective on the primary function of EVs, arguing that they serve as essential elements providing membrane area for long-distance, contact-dependent cellular communication based on protein-protein interaction. While EVs have been recognized as carriers of genetic information, additional unique advantages that they could provide for cellular communication remain unclear. Here, we introduce the concept that the substantial membrane area provided by EVs allows for membrane contact-dependent interactions that could be central to their function. This membrane area enables the lateral diffusion and sorting of membrane ligands like proteins, polysaccharides or lipids in two dimensions, promoting avidity-driven effects and assembly of co-stimulatory architectures at the EV-cell interface. The concept of vesicle-induced receptor sequestration (VIRS), for example, describes how EVs confine and focus receptors at the EV contact site, promoting a dense local concentration of receptors into signalosomes. This process can increase the signalling strength of EV-presented ligands by 10-1000-fold compared to their soluble counterparts. The speculations in this perspective advance our understanding of EV-biology and have critical implications for EV-based applications and therapeutics. We suggest a shift in perspective from viewing EVs merely as transporters of relevant nucleic acids and proteins to considering their unique biophysical properties as presentation platforms for long-distance, contact-dependent signalling. We therefore highlight the functional role of the EV membrane rather than their content. We further discuss how this signalling mechanism might be exploited by virus-transformed or cancer cells to enhance immune-evasive mechanisms.

Hakami, Niki | Burgstaller, Anna | Gao, Ning | Rutz, Angela | Mann, Stephen | Staufer, Oskar
DOI:
Microfluidics plays a pivotal role in organ-on-chip technologies and in the study of synthetic cells, especially in the development and analysis of artificial cell models. However, approaches that use synthetic cells as integral functional components for microfluidic systems to shape the microenvironment of natural living cells cultured on-chip are not explored. Here, colloidosome-based synthetic cells are integrated into 3D microfluidic devices, pioneering the concept of synthetic cell-based microenvironments for organs-on-chip. Methods are devised to create dense and stable networks of silica colloidosomes, enveloped by supported lipid bilayers, within microfluidic channels. These networks promote receptor-ligand interactions with on-chip cultured cells. Furthermore, a technique is introduced for the controlled release of growth factors from the synthetic cells into the channels, using a calcium alginate-based hydrogel formation within the colloidosomes. To demonstrate the potential of the technology, a modular plug-and-play lymph-node-on-a-chip prototype that guides the expansion of primary human T cells by stimulating receptor ligands on the T cells and modulating their cytokine environment is presented. This integration of synthetic cells into microfluidic systems offers a new direction for organ-on-chip technologies and suggests further avenues for exploration in potential therapeutic applications.
Welsh, Joshua A. | Goberdhan, Deborah C. I. | O'Driscoll, Lorraine | Buzas, Edit I. | Blenkiron, Cherie | Bussolati, Benedetta | Cai, Houjian | Di Vizio, Dolores | Driedonks, Tom A. P. | Erdbrügger, Uta | Falcon-Perez, Juan M. | Fu, Qing-Ling | Hill, Andrew F. | Lenassi, Metka | Lim, Sai Kiang | Mahoney, Mỹ G. | Mohanty, Sujata | Möller, Andreas | Nieuwland, Rienk | Ochiya, Takahiro | Sahoo, Susmita | Torrecilhas, Ana C. | Zheng, Lei | Zijlstra, Andries | Abuelreich, Sarah | Bagabas, Reem | Bergese, Paolo | Bridges, Esther M. | Brucale, Marco | Burger, Dylan | Carney, Randy P. | Cocucci, Emanuele | Crescitelli, Rossella | Hanser, Edveena | Harris, Adrian L. | Haughey, Norman J. | Hendrix, An | Ivanov, Alexander R. | Jovanovic-Talisman, Tijana | Kruh-Garcia, Nicole A. | Ku'ulei-Lyn Faustino, Vroniqa | Kyburz, Diego | Lässer, Cecilia | Lennon, Kathleen M. | Lötvall, Jan | Maddox, Adam L. | Martens-Uzunova, Elena S. | Mizenko, Rachel R. | Newman, Lauren A. | Ridolfi, Andrea | Rohde, Eva | Rojalin, Tatu | Rowland, Andrew | Saftics, Andras | Sandau, Ursula S. | Saugstad, Julie A. | Shekari, Faezeh | Swift, Simon | Ter-Ovanesyan, Dmitry | Tosar, Juan P. | Useckaite, Zivile | Valle, Francesco | Varga, Zoltan | van der Pol, Edwin | van Herwijnen, Martijn J. C. | Wauben, Marca H. M. | Wehman, Ann M. | Williams, Sarah | Zendrini, Andrea | Zimmerman, Alan J. | MISEV Consortium | Théry, Clotilde | Witwer, Kenneth W. | Staufer, Oskar
DOI:
Abstract Extracellular vesicles (EVs), through their complex cargo, can reflect the state of their cell of origin and change the functions and phenotypes of other cells. These features indicate strong biomarker and therapeutic potential and have generated broad interest, as evidenced by the steady year-on-year increase in the numbers of scientific publications about EVs. Important advances have been made in EV metrology and in understanding and applying EV biology. However, hurdles remain to realising the potential of EVs in domains ranging from basic biology to clinical applications due to challenges in EV nomenclature, separation from non-vesicular extracellular particles, characterisation and functional studies. To address the challenges and opportunities in this rapidly evolving field, the International Society for Extracellular Vesicles (ISEV) updates its ‘Minimal Information for Studies of Extracellular Vesicles’, which was first published in 2014 and then in 2018 as MISEV2014 and MISEV2018, respectively. The goal of the current document, MISEV2023, is to provide researchers with an updated snapshot of available approaches and their advantages and limitations for production, separation and characterisation of EVs from multiple sources, including cell culture, body fluids and solid tissues. In addition to presenting the latest state of the art in basic principles of EV research, this document also covers advanced techniques and approaches that are currently expanding the boundaries of the field. MISEV2023 also includes new sections on EV release and uptake and a brief discussion of in vivo approaches to study EVs. Compiling feedback from ISEV expert task forces and more than 1000 researchers, this document conveys the current state of EV research to facilitate robust scientific discoveries and move the field forward even more rapidly.

