Publications
Dey, Sourik | Sankaran, Shrikrishnan
DOI:
Recent advances in engineered bacterial therapeutics underscore their potential in treating diseases via targeted, live interventions. Despite their promising performance in early clinical phases, no engineered therapeutic bacteria have yet received approval, primarily due to challenges in proving efficacy while ensuring biosafety. Material science innovations, particularly the encapsulation of bacteria within hydrogels, present a promising avenue to enhance bacterial survival, efficacy, and safety in therapeutic applications. This review discusses this interdisciplinary approach to develop living therapeutic materials. Hydrogels not only safeguard the bacteria from harsh physiological conditions but also enable controlled therapeutic release and prevent unintended bacterial dissemination. The strategic use of encapsulation materials could redefine the delivery and functionality of engineered bacterial therapeutics, facilitating their clinical translation.
Dey, Sourik | Sankaran, Shrikrishnan
DOI:
Zhu et al. introduce MELG (materials engineered by living grafting), combining engineered microbes with non-living scaffolds for functional protein regeneration within. These MELGs can be used for long-term controlled release, enzyme-mediated biocatalysis, and DNA purification. This approach offers enhanced functionality and durability in bioactive materials compared to traditional non-living counterparts.
Dey, Sourik | Seyfert, Carsten E. | Fink-Straube, Claudia | Kany, Andreas Martin | Müller, Rolf | Sankaran, Shrikrishnan
DOI:
Peptide drugs have seen rapid advancement in biopharmaceutical development, with over 80 candidates approved
globally. Despite their therapeutic potential, the clinical translation of peptide drugs is hampered by challenges
in production yields and stability. Engineered bacterial therapeutics is a unique approach being explored to overcome
these issues by using bacteria to produce and deliver therapeutic compounds at the body site of use. A key advan‑
tage of this technology is the possibility to control drug delivery within the body in real time using genetic switches.
However, the performance of such genetic switches suffers when used to control drugs that require post‑translational
modifications or are toxic to the host. In this study, these challenges were experienced when attempting to establish
a thermal switch for the production of a ribosomally synthesized and post‑translationally modified peptide antibiotic,
darobactin, in probiotic E. coli. These challenges were overcome by developing a thermo‑amplifier circuit that com‑
bined the thermal switch with a T7 RNA Polymerase. Due to the orthogonality of the Polymerase, this strategy
overcame limitations imposed by the host transcriptional machinery. This circuit enabled production of pathogen‑
inhibitory levels of darobactin at 40 °C while maintaining leakiness below the detection limit at 37 °C. Furthermore,
the thermo‑amplifier circuit sustained gene expression beyond the thermal induction duration such that with only
2 h of induction, the bacteria were able to produce pathogen‑inhibitory levels of darobactin. This performance
was maintained even in physiologically relevant simulated conditions of the intestines that include bile salts and low
nutrient levels
Bhusari, Shardul | Kim, Juhyun | Polizzi, Karen | Sankaran, Shrikrishnan | del Campo, Aránzazu
DOI:
In engineered living materials (ELMs) non-living matrices encapsulate microorganisms to acquire capabilities like sensing or biosynthesis. The confinement of the organisms to the matrix and the prevention of overgrowth and escape during the lifetime of the material is necessary for the application of ELMs into real devices. In this study, a bilayer thin film hydrogel of Pluronic F127 and Pluronic F127 acrylate polymers supported on a solid substrate is introduced. The inner hydrogel layer contains genetically engineered bacteria and supports their growth, while the outer layer acts as an envelope and does not allow leakage of the living organisms outside of the film for at least 15 days. Due to the flat and transparent nature of the construct, the thin layer is suited for microscopy and spectroscopy-based analyses. The composition and properties of the inner and outer layer are adjusted independently to fulfil viability and confinement requirements. We demonstrate that bacterial growth and light-induced protein production are possible in the inner layer and their extent is influenced by the crosslinking degree of the used hydrogel. Bacteria inside the hydrogel are viable long term, they can act as lactate-sensors and remain active after storage in phosphate buffer at room temperature for at least 3 weeks. The versatility of bilayer bacteria thin-films is attractive for fundamental studies and for the development of application-oriented ELMs.
Basaran, Selim | Dey, Sourik | Bhusari, Shardul | Sankaran, Shrikrishnan | Kraus, Tobias
DOI:
Engineered living materials (ELMs) encapsulate microorganisms within polymeric matrices for biosensing, drug delivery, capturing viruses, and bioremediation. It is often desirable to control their function remotely and in real time and so the microorganisms are often genetically engineered to respond to external stimuli. Here, we combine thermogenetically engineered microorganisms with inorganic nanostructures to sensitize an ELM to near infrared light. For this, we use plasmonic gold nanorods (AuNR) that have a strong absorption maximum at 808 nm, a wavelength where human tissue is relatively transparent. These are combined with Pluronic-based hydrogel to generate a nanocomposite gel that can convert incident near infrared light into heat locally. We perform transient temperature measurements and find a photothermal conversion efficiency of 47 %. Steady-state temperature profiles from local photothermal heating are quantified using infrared photothermal imaging and correlated with measurements inside the gel to reconstruct spatial temperature profiles. Bilayer geometries are used to combine AuNR and bacteria-containing gel layers to mimic core-shell ELMs. The thermoplasmonic heating of an AuNR-containing hydrogel layer that is exposed to infrared light diffuses to the separate but connected hydrogel layer with bacteria and stimulates them to produce a fluorescent protein. By tuning the intensity of the incident light, it is possible to activate either the entire bacterial population or only a localized region.
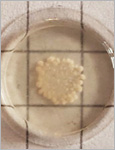
Riedel, Florian | Puertas Bartolomé, María | Teruel Enrico, Lara Luana | Fink-Straube, Claudia | Nguyen Dong, Cao | Gherlone, Fabio | Huang, Ying | Valiante, Vito | del Campo, Aránzazu | Sankaran, Shrikrishnan
DOI:
Microbial biofactories allow the upscaled production of high-value compounds in biotechnological processes. This is particularly advantageous for compounds like flavonoids that promote better health through their antioxidant, antibacterial, anticancer and other beneficial effects but are only produced in small quantities in their natural plant-based hosts. Bacteria like E. coli have been genetically modified with enzyme cascades to produce flavonoids like naringenin and pinocembrin from coumaric or cinnamic acid. Despite advancements in yield optimization, the production of these compounds still involves high costs associated with their biosynthesis, purification, storage and transport. An alternative production strategy could involve the direct delivery of the microbial biofactories to the body. In such a strategy, ensuring biocontainment of the engineered microbes in the body and controlling production rates are major challenges. In this study, these two aspects are addressed by developing engineered living materials (ELMs) consisting of probiotic microbial biofactories encapsulated in biocompatible hydrogels. Engineered probiotic E. coli Nissle 1917 able to efficiently convert cinnamic acid into pinocembrin were encapsulated in poly(vinyl alcohol)-based hydrogels. The biofactories are contained in the hydrogels for a month and remain metabolically active during this time. Control over production levels is achieved by the containment inside the material, which regulates bacteria growth, and by the amount of cinnamic acid in the medium.
Blanch-Asensio, Marc | Dey, Sourik | Sankaran, Shrikrishnan
DOI:
Lactobacilli are gram-positive bacteria that are growing in importance for the healthcare industry and genetically engineering them as living therapeutics is highly sought after. However, progress in this field is hindered since most strains are difficult to genetically manipulate, partly due to their complex and thick cell walls limiting our capability to transform them with exogenous DNA. To overcome this, large amounts of DNA (>1 µg) are normally required to successfully transform these bacteria. An intermediate host, like E. coli, is often used to amplify recombinant DNA to such amounts although this approach poses unwanted drawbacks such as an increase in plasmid size, different methylation patterns and the limitation of introducing only genes compatible with the intermediate host. In this work, we have developed a direct cloning method based on in-vitro assembly and PCR amplification to yield recombinant DNA in significant quantities for successful transformation in L. plantarum WCFS1. The advantage of this method is demonstrated in terms of shorter experimental duration and the possibility to introduce a gene incompatible with E. coli into L. plantarum WCFS1.

Dey, Sourik | Blanch-Asensio, Marc | Kuttae, Sanjana Balaji | Sankaran, Shrikrishnan
DOI:
Lactobacilli are ubiquitous in nature, often beneficially associated with animals as commensals and probiotics, and are extensively used in food fermentation. Due to this close-knit association, there is considerable interest to engineer them for healthcare applications in both humans and animals, for which high-performance and versatile genetic parts are greatly desired. For the first time, we describe two genetic modules in Lactiplantibacillus plantarum that achieve high-level gene expression using plasmids that can be retained without antibiotics, bacteriocins or genomic manipulations. These include (i) a promoter, PtlpA, from a phylogenetically distant bacterium, Salmonella typhimurium, which drives up to 5-fold higher level of gene expression compared to previously reported promoters and (ii) multiple toxin-antitoxin systems as a self-contained and easy-to-implement plasmid retention strategy that facilitates the engineering of tuneable transient genetically modified organisms. These modules and the fundamental factors underlying their functionality that are described in this work will greatly contribute to expanding the genetic programmability of lactobacilli for healthcare applications.

Dhakane, Priyanka | Tadimarri, Varun S. | Sankaran, Shrikrishnan
DOI:
Regenerative medicine aims to restore damaged cells, tissues, and organs, for which growth factors are vital to stimulate regenerative cellular transformations. Major advances have been made in growth factor engineering and delivery like the development of robust peptidomimetics and controlled release matrices. However, their clinical applicability remains limited due to their poor stability in the body and need for careful regulation of their local concentration to avoid unwanted side-effects. In this study, a strategy to overcome these limitations is explored using engineered living materials (ELMs), which contain live microorganisms that can be programmed with stimuli-responsive functionalities. Specifically, the development of an ELM that releases a pro-angiogenic protein in a light-regulated manner is described. This is achieved by optogenetically engineering bacteria to synthesize and secrete a vascular endothelial growth factor peptidomimetic (QK) linked to a collagen-binding domain. The bacteria are securely encapsulated in bilayer hydrogel constructs that support bacterial functionality but prevent their escape from the ELM. In situ control over the release profiles of the pro-angiogenic protein using light is demonstrated. Finally, it is shown that the released protein is able to bind collagen and promote angiogenic network formation among vascular endothelial cells, indicating the regenerative potential of these ELMs.

