Building Immunity from the Bottom Up
The Emmy Noether Research Group Immuno Materials develops new synthetic cell-based approaches to regulate and study immune reactions. By applying model membrane systems and in vitro reconstitution approaches, inspired from bottom-up synthetic biology, we engineer controlled cellular environments that trigger immune reaction in T cells and cancer cells. With this approach, we study how biophysical and biochemical cues guide cancer-immune signaling and apply this knowledge to improve next generation immunotherapies.

Mitarbeiter/innen



Research
Synthetic tumor immune microenvironments
Major research questions
How do cancer cells adapt to their immune microenvironment? How does the presence of immune cells around cancer cells affect their response to therapy? How do the biophysical properties of tumors change when immune cell infiltrate into them?
Within the framework of this project, we develop artificial tumour immune microenvironments (TIMEs) for human pancreatic cancer organoids empowering structured and rational analysis of tumour immune adaptations Immune cells, the defining elements of an immune microenvironment are recreated as synthetic cells by bottom-up assembly from their single molecular building blocks The programmable synthetic cells are introduced into tumour organoids to function as lifelike leukocyte mimics presenting immune effector functions By this, a molecularly defined immune environment is created inside tumour models Multi-parametric screenings assess organoid development as well as immunotherapy response as a function of the synthetic microenvironment configuration This strategy links TIME architectures to cancer immune adaptation and evasion for quantitative description of therapy resistance The project strives to de-convolute the dynamic complexity of the tumour immune microenvironment towards a rational dissection Moreover, it contributes concepts for the assembly of hybrid biomaterials that embody essential features of living cells.
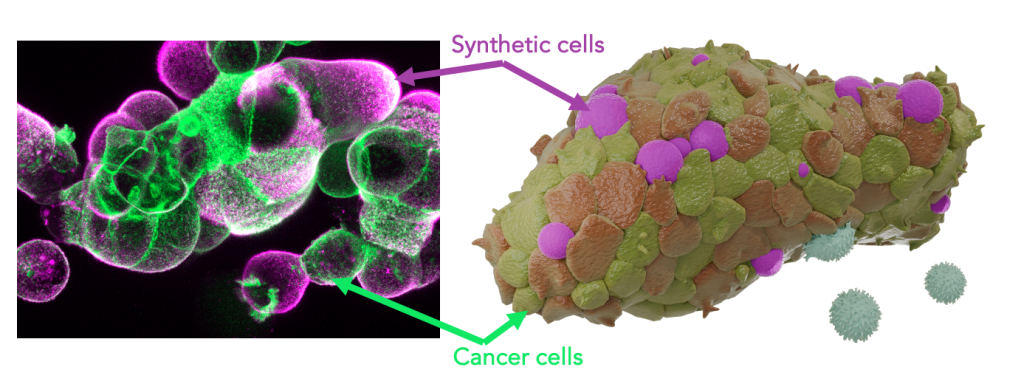
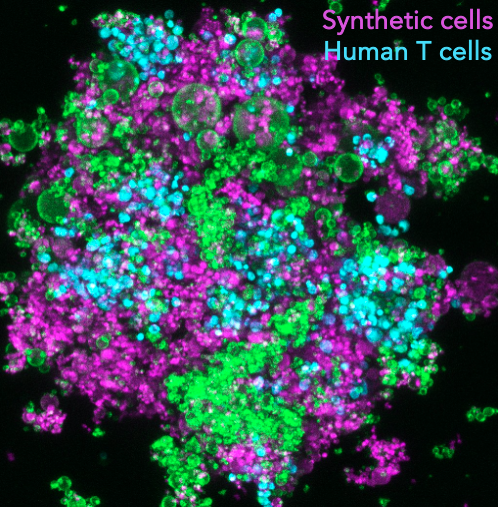
Synthetic lymphatic tissues
Major research questions
What are the microanatomical architectures and mechanical properties that favor T-cell stimulation in lymph nodes? What is the cross-correlation between mechanical and biochemical cues during T-cell activation? How do T cells collect and integrate signals within lymph nodes? How can T-cells be activated in a programmable, long-lasting and controlled way for therapeutic purposes?
T cells are instrumental in combatting diseases and fostering lasting immunity. Their maturation is a meticulously orchestrated process that takes place within lymph nodes, the central regulatory hubs for immunity. These nodes not only determine the stimulation of specific T cell types but also provide the optimal environment for their activation, ensuring a targeted and potent response. This maturation involves a sequence of events, including initial stimulation, monoclonal expansion, and differentiation. The sequence’s order and duration are vital for cultivating a diverse T cell population and a successful adaptive immune response. As T cells mature, the cellular microenvironment within the lymph nodes continually evolves, leading to changes in the biochemical and biomechanical properties of adjacent cells. Such alterations, like shifts in tissue stiffness or the introduction of various stimulatory ligands, guide T cell maturation and support their differentiation. For example, during T cell expansion, the lymph node environment sees about a 3-fold increase in stiffness, and nearby cells like antigen-presenting cells (APCs) or fibroblastic reticular cells (FRCs) release more stimulatory ligands and cytokines, such as IL-7 and CCL21. However, current in vitro T-cell expansion technologies fall short in replicating this intricate sequence of events. Our project aims to establish ex vivo T-cell expansion pipelines that mirror these in vivo processes, incorporating modules that adjust T-cell microenvironments using synthetic cells. This approach aims to boost the production of high-quality, therapeutic T cells for immunotherapy.
Synthetic tumor niches
Major research questions
How can leukemia cells hide in lymph nodes? Which survival and resistance signals do they receive there? How can we design ex vivo systems to mimic and study the relevant processes?
Leukemia, a form of blood cancer, remains a formidable challenge in oncology. Despite many patients initially responding to treatments and the advent of advanced immunotherapies that have notably enhanced prognoses, relapses remain a significant concern. This is largely attributed to certain leukemia cells, particularly cancer stem cells, finding sanctuary within lymph nodes. These nodes, under normal physiological conditions, offer a nourishing environment for white blood cells to proliferate and thrive. However, under pathological conditions, such as in cancer, they can inadvertently provide a protective haven for therapy-resistant leukemia cells. In this project, we aim to decipher the key signals lymph nodes extend to resident leukemia cells that confer therapy resistance. To achieve this, we build our own synthetic lymph nodes from the bottom-up, controlling their biochemical and biophysical properties aligned with those observed in leukemia patients. Subsequent treatments with chemotherapy and antibody immunotherapies allow us to evaluate how the lymph node environment potentially aids cancer cell survival. This endeavor, while rooted in fundamental science, holds promise for offering predictive insights in a translational context.

Research Philosophy
In the ever-evolving landscape of scientific research, our lab stands firm on certain foundational beliefs that guide our inquiries and innovations. These principles not only shape our approach but also define our commitment to advancing the frontiers of knowledge.
- Balancing Fundamental Science with Technological Advances: At the heart of our work is a deep respect for fundamental science, understanding its role as the catalyst for technological innovations. Conversely, we recognize that new technologies can illuminate and deepen our fundamental insights. In this symbiotic relationship, our lab harnesses both the foundational and the innovative aspects of the life sciences.
- Synthetic Biology & Understanding by Design: Merely observing and perturbing molecules, cells, and organisms isn’t enough to truly grasp the intricacies of biological systems. We embrace the principles of synthetic biology, advocating for an „understand-by-design“ approach. By adopting an engineering lens towards life, we seek to derive quantitative insights and fortify our scientific discourse with new empirical evidence.
- Insight Over Data: While data-driven research forms the bedrock of science, we believe that conceptual insights often hold greater value. We believe in the power of ideas and emphasizing the significance of arguments is rooted in experiments and mechanisms over mere data accumulation.
Beyond these foundational pillars, our lab emphasizes inclusivity and collaboration. We foster an environment where individuals, regardless of their career stage, are encouraged to share ideas and constructive critiques. Open dialogue is our strength; it’s through candid and sometimes challenging discussions that we unearth groundbreaking insights and concepts. Our ultimate goal is to cultivate a nurturing space where students flourish and scientists are equipped with all they need to discover new perspectives.
Success stories
Though our lab was only recently established, we’ve been at the forefront of pioneering new concepts in synthetic biology and immunotherapy. Our contributions have already made significant impacts.
Beyond the Spike: Pioneering Discoveries in the Heart of a Pandemic
The COVID-19 pandemic tested us all, both personally and professionally. As nations grappled with lockdowns, scientists globally ramped up their efforts to decode the mysteries of the SARS-CoV-2 virus. We all played our part.
In the summer of 2020, at the pandemic’s onset, we collaborated with an interdisciplinary team from the University of Bristol and the Max Planck Center Bristol in the UK. Our colleagues there achieved a groundbreaking discovery: they identified the molecular structure of the SARS-CoV-2 spike protein binding unexpectedly to a fatty acid, specifically linoleic acid (Toelzer et al. (2020) Science). This finding was monumental. The fatty acid essentially „locked“ the spike protein, preventing it from infecting cells. We soon recognized the profound implications this could have on our understanding of COVID-19.
As the pandemic evolved, we delved deeper into the fatty acid binding pocket’s role. Notably, all emerging SARS-CoV-2 variants retained this binding pocket. Using our understand-by-design approach, in partnership with the Max Planck Center, we crafted non-infective, molecularly defined minimal synthetic variants of SARS-CoV-2 virions. This allowed us to methodically analyze the fatty acid’s role in virus-cell docking (Staufer et al. (2021) Nature Communications). Our research led to a model that elucidated the evolutionary advantage of the fatty acid binding pocket: it shields the virus from immune detection. This molecular switch modulates the virus’s infectivity, adapting to the immune system’s local activity and inflammation.
Our minimal virions also shed light on the evolution and diversity of SARS-CoV-2 (Gupta et al. (2021) Nature Communications) and provided insights into how the virus impedes the formation of immune synapses (Onnis et al. (2022) Journal of Experimental Medicine). Our discoveries surrounding the fatty acid binding pocket even spurred the creation of Halo Therapeutics, a UK-based company now developing pan-coronavirus treatments. Halo just completed its Series A funding!
In summary, our journey has been both challenging and rewarding. We’ve made groundbreaking scientific discoveries, engaged in enriching discussions, and forged new partnerships, all underpinned by our understand-by-design approach. It’s a testament to the power of collaboration and innovation in the face of adversity.
Synthetic Surge: Engineering Life’s Blueprint in the New Age of Biology
In the realm of biology, the understand-by-design approach isn’t a newcomer. For decades, scientists have endeavored to recreate cellular life. Yet, it’s the emergence of advanced molecular systems engineering and the capability to craft supramolecular systems in test tubes that has truly transformed our pace. Now, we can swiftly replicate natural cells with varied functions, from cell division to regulating cellular immunity.
Armed with these cutting-edge tools, the global race towards synthetic cells is in full swing. Numerous labs and initiatives are sprouting, all with a shared vision: to birth the first living synthetic cells. While some delve deep into life’s fundamentals, others see a future where these synthetic cells, being more controllable, can revolutionize sustainability and medicine.
Spurred by the SynCell conference in 2021 and in collaboration with global partners, we’ve embarked on a mission to streamline these worldwide endeavors. Joining forces with early-career scientists from nations like the US, UK, Netherlands, Spain, Germany, France, Japan, and Australia, we’ve brainstormed ways to attract the next generation of innovators to this field. Our aspiration goes beyond just cells; we aim to build a community to engineer synthetic cells and organelles from the bottom-up! We’ve strategized to amplify the socioeconomic and technological impacts of synthetic cells and emphasized the importance of an interconnected, interdisciplinary research community (Staufer et al. (2021) eLife). By bolstering communication, sharing resources, and initiating educational programs, we believe in a sustainable future for this field.
Our journey, bridging continents and disciplines, stands as a testament to the collaborative spirit of science. Witnessing the passion and drive of early-career scientists has been turely enriching!
Open position
We pride ourselves on being a diverse team, both culturally and scientifically. Our expertise spans a wide range of the natural sciences, including biophysics, immunology, advanced microscopy, microfluidics, nanotechnology, drug delivery, immunotherapy, virology, molecular biology, membranes, electrophysiology, cell biology, physical chemistry, and more. We invite you to reach out and become a part of our vibrant community.
Postdoctoral Researchers: We have fully-funded postdoc positions available. If you’re considering fellowships, we provide guidance and support for applications, including but not limited to the Marie Curie Fellowship, DFG Walter Benjamin Program, and the Feodor Lynen Fellowship by the Humboldt Foundation. We value your unique research ideas for other programs and offer constructive discussions within our group to help you craft a successful proposal. For postdoc opportunities, please get in touch by sending your interest statement to us directly.
PhD Candidates: We invite applications for our fully funded PhD positions. Our research projects are co-defined with students, emphasizing interdisciplinary training and state-of-the-art methods spanning biophysics, microfluidics, immunology, advanced microscopy, and 3D cell culturing. Send us your motivation letter and CV. We’re eager to hear about your research ideas and welcome you to explore them in our lab.
Master’s Students: We offer opportunities for master’s thesis projects in areas like synthetic cells, immunotherapy, organoids, and biomaterials. While these subjects have a predefined framework, we encourage students to add their unique perspective. Every master’s student is mentored directly by an experienced PhD candidate or postdoc.
High School Students: Unsure about your academic path? Curious about the dynamics of a research lab? We offer short-term research internships, providing a glimpse into the world of natural sciences. Join us and get a hands-on experience.
Publikationen

Burgstaller, Anna | Nink, Tamara | Walter, Niklas | Lopez Lopez, Erick Angel | Chang, Shin-Fang | Staufer, Oskar
DOI:
Synthetic cells have emerged as a novel biomimetic approach for studying fundamental cellular functions and enabling new therapeutic interventions. However, the potential to program synthetic cells into self-organized 3D collectives to replicate the structure and function of tissues has remained largely untapped. Here, self-assembly properties are engineered into synthetic cells to form millimeter-sized 3D lymphatic bottom-up tissues (lymphBUTs) with mechanical adaptability, metabolic activity, and hierarchical microstructural organization. It is demonstrated that primary human immune cells spontaneously infiltrate and functionally integrate into these synthetic lymph nodes to form living tissue hybrids. Applying lymphBUTs, it is shown that structured 3D organization and mechanical support drives T cell activation and the application of lymphBUTs for ex vivo expansion of regulatory CD8+ T cells is demonstrated. The study highlights the functional integration of living and non-living matter, advancing synthetic cell engineering toward 3D tissue structures.
Zhou, Xiangda | Zhang, Sijia | Yang, Wenjuan | Gonder, Susanne | Sadjadi, Zeinab | Piernitzki, Nils | Moter, Alina | Sharma, Shulagna | Largeot, Anne | Küchler, Nadja | Kaschek, Lea | Schäfer, Gertrud | Schwarz, Eva C. | Eichler, Hermann | Ullrich, Evelyn | Rieger, Heiko | Staufer, Oskar | Paggetti, Jérome | Moussay, Etienne | Hoth, Markus | Qu, Bin
DOI:
Natural killer (NK) cells are critical components of the first-line immune defense, responsible for eliminating tumorigenic cells. NK cell-based adoptive immunotherapy has gained increasing attention; however, cryopreservation, a standard technique for NK cell storage, significantly impairs NK cell cytotoxicity, particularly in physiological 3D environments. Here, we demonstrate that short-term co-culture with effector T cells markedly enhances NK cell motility and killing functionality. Notably, a brief 1-day co-culture is sufficient to restore cryopreservation-impaired NK cell functionality in 3D environments. This enhancement requires direct contact between T cells and NK cells, which facilitates localized high concentrations of IL-2 at the cell contact sites. To develop a controled, donor-independent solution, we demonstrate that synthetic T cells with surface-bound IL-2 exhibit superior efficiency in revitalizing cryopreserved NK cells. These findings uncover a previously unrecognized role for physical contact-mediated local IL-2 signaling and provide an efficient, cost-effective, and tunable strategy to rescue NK cell functionality post-cryopreservation, paving the way for more scalable, potent, and clinically viable NK cell-based immunotherapies.
Leithner, Alexander | Staufer, Oskar | Mitra, Tanmay | Liberta, Falk | Valvo, Salvatore | Kutuzov, Mikhail | Dada, Hannah | Spaeth, Jacob | Zhou, Weijie | Schiele, Felix | Reindl, Sophia | Nar, Herbert | Hoerer, Stefan | Crames, Maureen | Comeau, Stephen | Young, David | Low, Sarah | Jenkins, Edward | Davis, Simon J. | Klenerman, David | Nixon, Andrew | Pefaur, Noah | Wyatt, David | Dushek, Omer | Kasturirangan, Srinath | Dustin, Michael L.
DOI:
Bispecific T cell engagers (TcEs) link T cell receptors to tumor-associated antigens on cancer cells, forming cytotoxic immunological synapses (IS). Close membrane-to-membrane contact (≤13 nm) has been proposed as a key mechanism of TcE function. To investigate this and identify potential additional mechanisms, we compared four immunoglobulin G1-based (IgG1) TcE Formats (A–D) targeting CD3ε and Her2, designed to create varying intermembrane distances (A < B < C < D). Small-angle X-ray scattering (SAXS) and modeling of the conformational states of isolated TcEs and TcE–antigen complexes predicted close contacts (≤13 nm) for Formats A and B and far contacts (≥18 nm) for Formats C and D. In supported lipid bilayer (SLB) model interfaces, Formats A and B recruited, whereas Formats C and D repelled, CD2–CD58 interactions. Formats A and B also excluded bulky Quantum dots more effectively. SAXS also revealed that TcE–antigen complexes formed by Formats A and C were less flexible than complexes formed by Formats B and D. Functional data with Her2-expressing tumor cells showed cytotoxicity, surface marker expression, and cytokine release following the order A > B = C > D. In a minimal system for IS formation on SLBs, TcE performance followed the trend A = B = C > D. Addition of close contact requiring CD58 costimulation revealed phospholipase C-γ activation matching cytotoxicity with A > B = C > D. Our findings suggest that when adhesion is equivalent, TcE potency is determined by two parameters: contact distance and flexibility. Both the close/far-contact formation axis and the low/ high flexibility axis significantly impact TcE potency, explaining the similar potency of Format B (close contact/high flexibility) and C (far contact/low flexibility). Copyright © 2025 the Author(s).
Burgstaller, Anna | Madureira, Sara | Staufer, Oskar
DOI:
Tissue functions rely on complex structural, biochemical, and biomechanical cues that guide cellular behavior and organization. Synthetic cells, a promising new class of biomaterials, hold significant potential for mimicking these tissue properties using simplified, nonliving building blocks. Advanced synthetic cell models have already shown utility in biotechnology and immunology, including applications in cancer targeting and antigen presentation. Recent bottom-up approaches have also enabled synthetic cells to assemble into 3D structures with controlled intercellular interactions, creating tissue-like architectures. Despite these advancements, challenges remain in replicating multicellular behaviors and dynamic mechanical environments. Here, we review recent advancements in synthetic cell-based tissue formation and introduce a three-pillar framework to streamline the development of synthetic tissues. This approach, focusing on synthetic extracellular matrix integration, synthetic cell self-organization, and adaptive biomechanics, could enable scalable synthetic tissues engineering for regenerative medicine and drug development.
Piernitzki, Nils | Gao, Ning | Gasparoni, Gilles | Krauß, Louisa M. | Schulze-Hentrisch, Julia | Dustin, Michael | Schrul, Bianca | Györffy, Balázs | Mann, Stephen | Staufer, Oskar
DOI:
Self-assembly is a fundamental property of living matter that drives the three-dimensional organization of cell collectives such as tissues and organs. Here, the co-assembly of synthetic and natural cells is leveraged to create hybrid living 3D cancer cultures. We screen a range of synthetic cell models for their ability to form augmented tumoroids with artificial but controllable micro-environments, and show that the balance of inter- and extracellular adhesion and synthetic cell surface tension are key material properties driving integrated co-assembly. We demonstrate that synthetic cells based on droplet-supported lipid bilayers can establish artificial tumor immune microenvironments (ART-TIMEs), mimicking immunogenic signals within tumoroids and eliminating the need to integrate complex living immune cells. Using the ART-TIME approach, we identify a AhR-ARNT-mediated co-signaling mechanism between PD-1 and CD2 as a driver in immune evasion of pancreatic ductal adenocarcinoma. Our study advances the field of hybrid organoid engineering, offers opportunities for the construction and modelling of artificial tumour environments, and marks a step towards the design of functional living/non-living cytomimetic materials.
Piernitzki, Nils | Staufer, Oskar
DOI:
Extracellular vesicles (EVs) are lipid-membrane-enclosed particles released from cells, playing a pivotal role in cellular communication, particularly within the immune system. The fundamental molecular mechanisms through which EVs offer unique functionality for immunotherapeutic benefits are identified and reviewed. The focus is on three essential features, all rooted in the EV lipid membrane: immune receptor–ligand interactions at the EV membrane interface, the shielding of immunogenic cargo within the EVs, and the fusion of EVs with target cell membranes for direct cargo delivery. From this, how these distinct EV attributes, from their initial description and analysis in immune communication, have led to the development of novel immunotherapeutic strategies is traced. This review delves into how these strategies are applied in various immunotherapies, such as cancer immunotherapy, autoimmune diseases, infections, vaccinations, and graft-versus-host diseases, to modulate communication among different cell types for immune regulation. It is concluded by reviewing clinical trials involving EVs in immunotherapy that have effectively harnessed EVs' unique molecular mechanisms in clinical settings. Research and standardization efforts to maximize the potential impact of EVs on immunotherapy are further suggested.
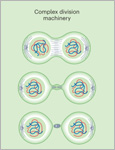
DOI:
In a major advancement for synthetic biology, dynamin A has been identified as a minimal component enabling cell division in synthetic cells, moving us one step nearer to realizing the ambition of creating synthetic life forms.
Jahnke, Kevin | Staufer, Oskar
DOI:
Extracellular vesicles (EVs), lipid-enclosed structures released by virtually all life forms, have gained significant attention due to their role in intercellular and interorganismal communication. Despite their recognized importance in disease processes and therapeutic applications, fundamental questions about their primary function remain. Here, we propose a different perspective on the primary function of EVs, arguing that they serve as essential elements providing membrane area for long-distance, contact-dependent cellular communication based on protein-protein interaction. While EVs have been recognized as carriers of genetic information, additional unique advantages that they could provide for cellular communication remain unclear. Here, we introduce the concept that the substantial membrane area provided by EVs allows for membrane contact-dependent interactions that could be central to their function. This membrane area enables the lateral diffusion and sorting of membrane ligands like proteins, polysaccharides or lipids in two dimensions, promoting avidity-driven effects and assembly of co-stimulatory architectures at the EV-cell interface. The concept of vesicle-induced receptor sequestration (VIRS), for example, describes how EVs confine and focus receptors at the EV contact site, promoting a dense local concentration of receptors into signalosomes. This process can increase the signalling strength of EV-presented ligands by 10-1000-fold compared to their soluble counterparts. The speculations in this perspective advance our understanding of EV-biology and have critical implications for EV-based applications and therapeutics. We suggest a shift in perspective from viewing EVs merely as transporters of relevant nucleic acids and proteins to considering their unique biophysical properties as presentation platforms for long-distance, contact-dependent signalling. We therefore highlight the functional role of the EV membrane rather than their content. We further discuss how this signalling mechanism might be exploited by virus-transformed or cancer cells to enhance immune-evasive mechanisms.

Hakami, Niki | Burgstaller, Anna | Gao, Ning | Rutz, Angela | Mann, Stephen | Staufer, Oskar
DOI:
Microfluidics plays a pivotal role in organ-on-chip technologies and in the study of synthetic cells, especially in the development and analysis of artificial cell models. However, approaches that use synthetic cells as integral functional components for microfluidic systems to shape the microenvironment of natural living cells cultured on-chip are not explored. Here, colloidosome-based synthetic cells are integrated into 3D microfluidic devices, pioneering the concept of synthetic cell-based microenvironments for organs-on-chip. Methods are devised to create dense and stable networks of silica colloidosomes, enveloped by supported lipid bilayers, within microfluidic channels. These networks promote receptor-ligand interactions with on-chip cultured cells. Furthermore, a technique is introduced for the controlled release of growth factors from the synthetic cells into the channels, using a calcium alginate-based hydrogel formation within the colloidosomes. To demonstrate the potential of the technology, a modular plug-and-play lymph-node-on-a-chip prototype that guides the expansion of primary human T cells by stimulating receptor ligands on the T cells and modulating their cytokine environment is presented. This integration of synthetic cells into microfluidic systems offers a new direction for organ-on-chip technologies and suggests further avenues for exploration in potential therapeutic applications.
Welsh, Joshua A. | Goberdhan, Deborah C. I. | O’Driscoll, Lorraine | Buzas, Edit I. | Blenkiron, Cherie | Bussolati, Benedetta | Cai, Houjian | Di Vizio, Dolores | Driedonks, Tom A. P. | Erdbrügger, Uta | Falcon-Perez, Juan M. | Fu, Qing-Ling | Hill, Andrew F. | Lenassi, Metka | Lim, Sai Kiang | Mahoney, Mỹ G. | Mohanty, Sujata | Möller, Andreas | Nieuwland, Rienk | Ochiya, Takahiro | Sahoo, Susmita | Torrecilhas, Ana C. | Zheng, Lei | Zijlstra, Andries | Abuelreich, Sarah | Bagabas, Reem | Bergese, Paolo | Bridges, Esther M. | Brucale, Marco | Burger, Dylan | Carney, Randy P. | Cocucci, Emanuele | Crescitelli, Rossella | Hanser, Edveena | Harris, Adrian L. | Haughey, Norman J. | Hendrix, An | Ivanov, Alexander R. | Jovanovic-Talisman, Tijana | Kruh-Garcia, Nicole A. | Ku’ulei-Lyn Faustino, Vroniqa | Kyburz, Diego | Lässer, Cecilia | Lennon, Kathleen M. | Lötvall, Jan | Maddox, Adam L. | Martens-Uzunova, Elena S. | Mizenko, Rachel R. | Newman, Lauren A. | Ridolfi, Andrea | Rohde, Eva | Rojalin, Tatu | Rowland, Andrew | Saftics, Andras | Sandau, Ursula S. | Saugstad, Julie A. | Shekari, Faezeh | Swift, Simon | Ter-Ovanesyan, Dmitry | Tosar, Juan P. | Useckaite, Zivile | Valle, Francesco | Varga, Zoltan | van der Pol, Edwin | van Herwijnen, Martijn J. C. | Wauben, Marca H. M. | Wehman, Ann M. | Williams, Sarah | Zendrini, Andrea | Zimmerman, Alan J. | MISEV Consortium | Théry, Clotilde | Witwer, Kenneth W. | Staufer, Oskar
DOI:
Abstract Extracellular vesicles (EVs), through their complex cargo, can reflect the state of their cell of origin and change the functions and phenotypes of other cells. These features indicate strong biomarker and therapeutic potential and have generated broad interest, as evidenced by the steady year-on-year increase in the numbers of scientific publications about EVs. Important advances have been made in EV metrology and in understanding and applying EV biology. However, hurdles remain to realising the potential of EVs in domains ranging from basic biology to clinical applications due to challenges in EV nomenclature, separation from non-vesicular extracellular particles, characterisation and functional studies. To address the challenges and opportunities in this rapidly evolving field, the International Society for Extracellular Vesicles (ISEV) updates its ‘Minimal Information for Studies of Extracellular Vesicles’, which was first published in 2014 and then in 2018 as MISEV2014 and MISEV2018, respectively. The goal of the current document, MISEV2023, is to provide researchers with an updated snapshot of available approaches and their advantages and limitations for production, separation and characterisation of EVs from multiple sources, including cell culture, body fluids and solid tissues. In addition to presenting the latest state of the art in basic principles of EV research, this document also covers advanced techniques and approaches that are currently expanding the boundaries of the field. MISEV2023 also includes new sections on EV release and uptake and a brief discussion of in vivo approaches to study EVs. Compiling feedback from ISEV expert task forces and more than 1000 researchers, this document conveys the current state of EV research to facilitate robust scientific discoveries and move the field forward even more rapidly.


